PDF chapter test TRY NOW
In order to understand how strong is an acid or a base first, let us see the classification of acids and bases based on the following characteristics
- Strength
- Basicity\ Acidity
- Concentration
Acids:
Acids are substances that have increased \(H^+\) ion concentration in an aqueous solution. These are acidic in nature.
1. Strength:
- Strong acid: When complete ionisation of acids takes place in an aqueous solution, it is known as strong acids. Here the high concentration of \(H^+\) ions is present.
- Weak acid: When partial ionisation of acids takes place in an aqueous solution, it is known as weak acids. Here the low concentration of \(H^+\) ions is present.
2. Basicity: The number of ions (\(H^+\) or \(H_3O^+\) ) produced by the ionisation of one molecule of acid in an aqueous solution is known as its basicity.
- Monobasic acids: The production of one hydronium ion per molecule of acid is known as monobasic acids. For example, hydrochloric acid \(HCl\), hydrobromic acid \(HBr\), hydrogen iodide \(HI\), and acetic acid \(CH3COOH\).
\(CH_3COOH \rightleftharpoons CH_3COO^- + H^+\)
- Dibasic acids: The production of two hydronium ions per molecule of acid is known as monobasic acids. For example, sulphuric acid \(H_2SO_4\), carbonic acid \(H_2CO_3\), and oxalic acid \(H_2C_2O_4\)
\(H_2SO_4 + H_2O \rightleftharpoons H_3O^+ + {HSO}^-_4\)
\({HSO}^-_4 + H_2O \rightleftharpoons H_3O^+ + {SO}^{2-}_4\)
- Tribasic acids: The production of three hydronium ions per molecule of acid is known as tribasic acids. For example, phosphoric acid \(H_3PO_4\), citric acid and so on.
\(H_3PO_4 + H_2O \rightleftharpoons H_3O^+ + {H_2PO}^-_4\)
\({H_2PO}^-_4 + H_2O \rightleftharpoons H_3O^+ {HPO}^{2-}_4\)
\({HPO}^{2-}_4 + H_2O \rightleftharpoons H_3O^+ + {PO}^{3-}_4\)
3. Concentration:
- Concentrated acid: If the aqueous solution contains a high percentage of acid and a low percentage of water, it is said to be concentrated acid.
- Dilute acid: If the aqueous solution contains a high percentage of water and a low percentage of acid, it is said to be diluted acid.
Bases:
Bases are substances that have increased \(OH^-\) ion concentration in an aqueous solution. These are basic in nature.
1. Strength:
- Strong base: When complete ionisation of bases takes place in an aqueous solution, it is known as strong bases. Here the high concentration of \(OH^-\) ions is present.
- Weak base: When partial ionisation of bases takes place in an aqueous solution, it is known as weak bases. Here the low concentration of \(OH^-\) ions is present.
2. Acidity: The number of ions \(OH^-\) produced by the ionisation of one molecule of a base in an aqueous solution is known as its acidity.
- Monoacidic bases: The production of one hydroxyl ion per molecule of a base is known as mono-acidic bases. For example, sodium hydroxide \(NaOH\), ammonium hydroxide \(NH_4OH\), potassium hydroxide \(KOH\) and so on.
\(NaOH \rightleftharpoons Na^+ + OH^-\)
- Di-acidic bases: The production of two hydroxyl ions per molecule of a base is known as di-acidic bases. For example, hydroxides of calcium, copper and magnesium, etc.
\(Ca{OH}_2 \rightleftharpoons Ca^{2+} + 2OH^-\)
- Tri-acidic bases: The production of three hydroxyl ions per molecule of a base is known as tri-acidic bases.
\(Fe{OH}_3 \rightleftharpoons Fe^{3+} + 3OH^-\)
3. Concentration:
- Concentrated base: If the aqueous solution contains a high percentage of base and a low percentage of water, it is said to be the concentrated base.
- Dilute base: If the aqueous solution contains a high percentage of water and a low percentage of base, it is said to be the diluted base.
From the above classifications, we can now distinguish between the types of acids and bases based on their strength, concentration, etc.
We know the taste of acidic and basic substances. Can we taste all of these?
No, it is because acids are corrosive and also some of the strong bases. To confirm a solution to be acid, base or neutral, we use a pH scale to identify them.
No, it is because acids are corrosive and also some of the strong bases. To confirm a solution to be acid, base or neutral, we use a pH scale to identify them.
To find the type of acids or bases its strength, we need to know how to use indicators and pH scale.
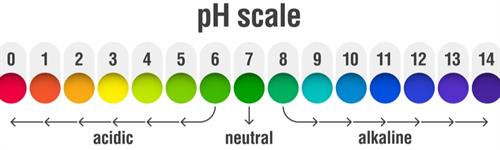
pH scale
Can we quantitatively find the number of ions present in a given solution?
Yes, we can with the help of a universal indicator which is actually a mixture of several indicators. A universal indicator shows different colours at different concentrations of hydrogen ions in a given solution.
Yes, we can with the help of a universal indicator which is actually a mixture of several indicators. A universal indicator shows different colours at different concentrations of hydrogen ions in a given solution.
The scale for measuring the hydrogen ion concentration in a solution is called the pH scale. Here, the 'p' stands for the German word potenz, meaning power. The number in the pH scale shows the pH value, which indicates the nature of the given solution.
The abbreviation for pH refers to the potential of hydrogen or the power of hydrogen. The pH scale ranges from \(0\) to \(14\), with \(0\) being the most acidic and \(14\) being the most alkaline. The pH of a solution determines whether it is acidic or alkaline.
Note: pH range \(7\) indicates the neutral solution.
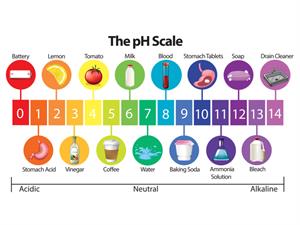
Different substances with pH range
Acids contain hydrogen ions \(H^+\), and thus lower pH value, whereas bases contain hydroxide ions \(OH^-\), and therefore higher pH value.
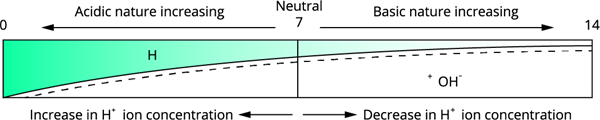
Variation of pH with the change in concentration of the \(H^+, OH^-\) ions.
The pH test is generally done by paper impregnated with the universal indicator for measuring the pH. This test determines how many hydrogen ions are present in a given solution.
A low pH (acidic substances) is produced by high hydrogen ion concentrations, while a high pH is produced by low hydrogen ion concentrations or high hydroxyl ion concentrations (basic substances).
A low pH (acidic substances) is produced by high hydrogen ion concentrations, while a high pH is produced by low hydrogen ion concentrations or high hydroxyl ion concentrations (basic substances).
Note: pH can also be written as \(pH= -log {[H]}^+\) which was given by S.P.L Sorensen
Example:
Let us now perform an activity to test the pH values of solutions.
Solutions:
- Saliva (before meal)
- Saliva (after meal)
- Lemon juice
- Colourless aerated drink
- Carrot juice
- Coffee
- Tomato juice
- Tap water
- \(1M NaOH\)
- \(1M HCl\)
Now record the observation and write down the nature of the substance based on it.
Sample solutions | Colour of pH paper | Approximate pH value | Nature of the substance |
Saliva (before meal) | Green | 7.4 | Basic |
Saliva (after meal) | Depends on the food | Depends on the food | Depends on the food |
Lemon juice | Pink | 2.5 | Acidic |
Colourless aerated drink | Yellow/Green | 5-6 | Acidic |
Carrot juice | Yellow/Green | 5-6 | Acidic |
Coffee | Yellow | 5 | Acidic |
Tomato juice | Orange | 4.1 | Acidic |
Tap water | Green | 6.5-8.5 | Neutral |
\(1M NaOH\) | Purple | 14 | Basic |
\(1M HCl\) | Red | 0 | Acidic |
Based on the observation, we can find the nature of the given solutions.
Now, let us perform a small activity to understand the strength of acids and bases:
Materials required:
- Hydrochloric acid
- Acetic acid
- Beakers
- Nails
- Cork
- Bulb
- Electric wire
Step 1: Take two beakers and name them A and B, respectively.
Step 2: Add hydrochloric acid in beaker A and acetic acid in beaker B, respectively.
Step 3: Fix two nails to a cork and connect it with a bulb through the wire. Both terminals are connected to a bulb with a \(6\) volt DC supply.
Step 2: Add hydrochloric acid in beaker A and acetic acid in beaker B, respectively.
Step 3: Fix two nails to a cork and connect it with a bulb through the wire. Both terminals are connected to a bulb with a \(6\) volt DC supply.
Step 4: Now, keep the cork connected to the bulb in each beaker separately.
Step 5: Allow the current to flow.
Observation: Bulb will glow brightly in a beaker with dilute \(HCl\), and the bulb's glow will be dimmer in a solution of dilute acetic acid.
Result: Electric current flows due to the presence of ions.
As we know, strong acids give rise to more \(H^+\) ions. At the same time, weak acids give rise to less number of \(H^+\) ions.
Result: Electric current flows due to the presence of ions.
Here, the dilute \(HCl\) has more ions making it a strong acid, whereas acetic acid has fewer ions, making it a week acid.
As we know, strong acids give rise to more \(H^+\) ions. At the same time, weak acids give rise to less number of \(H^+\) ions.
Therefore, we can conclude that we can determine the nature of given substances and their strength from the above experiments.
