
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoAn ionic compound is a pure substance composed of ions held together by electrostatic forces.
Example: \(NaCl\), \(LiCl\), \(CaCl_2\), \(CaO\), \(MgCl_2\)
Properties of ionic compounds:
Because of the strong force of attraction between the positive and negative ions, ionic compounds are solids but somewhat hard. When pressure is applied to such compounds, they become brittle and break into pieces.
Melting and boiling points:
The melting and boiling points of ionic compounds are very high. This is because breaking the strong inter-ionic interaction needs a lot of energy.
Solubility:
Electrovalent substances are soluble in water but insoluble in solvents such as kerosene, petrol, and other hydrocarbons.
Conduction of electricity:
The movement of charged particles is involved in the conduction of electricity through a solution. When electricity is passed through a solution containing an ionic substance in water, ions migrate to the opposite electrodes. Since ions cannot move in the solid-state due to their rigid structure, ionic compounds in this state cannot carry electricity.
However, in the molten state, ionic molecules conduct electricity. This is possible in the molten state because the heat overcomes the electrostatic forces of attraction between the oppositely charged ions. As a result, the ions are free to travel around and conduct electricity.
Let's check the conductivity of a salt solution with the activity below.
- Take a salt solution in a plastic container.
- Use LED instead of bulb since it glows even when a weak electric current flows through it.
- Make a battery (a couple of cells) and connect its positive terminal to the longer lead of the LED. Put the negative terminal of the battery into the container contains water.
- Put the shorter lead of the LED too into the salt solution using a wire.
- Make sure that both ends of the wire in the water do not touch and keep at least \(1\) \(cm\) distance.
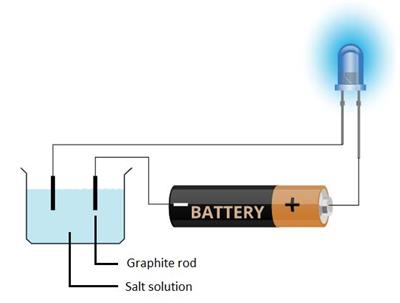
Testing conductivity of a salt solution
In this experiment, we can see that the LED glows that proves the salt solution is a good conductor of electricity.
Reference:
https://www.shutterstock.com/image-vector/collection-ion-flame-tests-zinc-potassiumstrontium-155936723
https://th.bing.com/th/id/OIP.x3vn84Aqx5JM0fBQ3dhSIwAAAA?pid=ImgDet&rs=1