
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoHuman beings are an essential part of the environment and are considered the most evolved species on earth. The activities performed by humans can affect and harm the environment around us. Our actions significantly impact the environment, which impacts humans, as we are an integral part of the environment.
Let us observe how the changes have affected us.
What is ozone?
Ozone (\(O_3\)) is a less stable and highly reactive molecule comprised of three oxygen atoms. While \(O_2\), commonly known as oxygen, is required for all aerobic forms of life. Ozone is a pale blue, pungent and poisonous gas.
Ozone, on the other hand, plays a vital role at higher altitudes in the atmosphere. The troposphere, stratosphere, mesosphere, thermosphere, and exosphere are the five layers of our atmosphere. About \(90 \%\) of the earth's ozone is concentrated in the stratosphere, the second layer of our atmosphere.

Ozone layer
Ozone in the stratosphere protects the earth's surface from the sun's damaging ultraviolet (UV) radiation. Ozone is present at \(23 - 25\) \(km\) above the equator and \(11 - 18\) \(km\) above the poles. This zone is called the ozone layer or ozonosphere. It is also called the ozone shield, as the region protects and intercepts the UV radiation.

Filtering of harmful radiations in the ozone layer
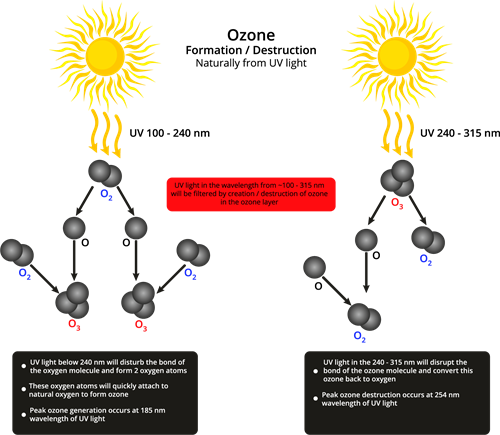
Formation of ozone layer:
At the stratosphere, the ozone is the product of UV radiation acting on the oxygen (\(O_2\)) molecule. UV light having higher energy splits the molecular oxygen (\(O_2\)) into free oxygen (\(O\)) atoms. These free oxygen combine with molecular oxygen (\(O_2\)) to form ozone.
\(O_2\) \(O\) \(+\) \(O\) (\(Free \ oxygen \ atom\))
\(O\) \(+\) \(O_2\) \(O_3\) (\(Ozone\))
Ozone layer formation
Important!
The United Nations General Assembly (UNGA) declared September \(16^{th}\), \(1994\), as World Ozone Day, an international day to preserve the ozone layer. The day signifies the signing of the Montreal Protocol on compounds that deplete the ozone layer in the year \(1987\) to protect and safeguard the ozone layer and prevent further damage.

World ozone day
Reference:
https://www.shutterstock.com/image-vector/ozone-formation-destruction-infographic-diagram-by-698017582
https://www.shutterstock.com/image-vector/world-ozone-day-creative-concept-2038746413