
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoCorrosion:
It is a gradual destruction of metals by chemical or electrochemical reactions to the environment. It is a natural process that converts metal into its oxide, hydroxide or sulphide to lose its metallic characteristics.

Natural corrosion
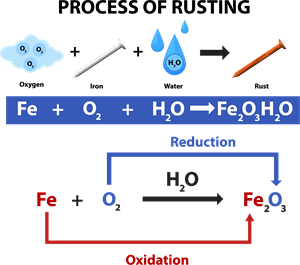
Rusting:
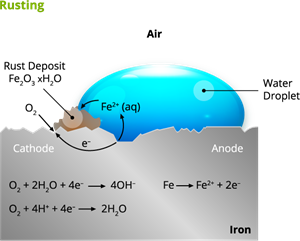
Rust is chemically known as hydrated ferric oxide (it is formulated as \(Fe_2O_3\). x\(H_2O\)). Rusting results in the formation of scaling reddish-brown hydrated ferric oxide on the surface of iron and iron-containing materials.
Corrosion types:
(i). Dry corrosion or Chemical corrosion:
Dry corrosion is the corrosive action that occurs in the absence of moisture. It is the chemical attack on a metal by corrosive liquids or gases such as \(O_2\), \(N_2\), \(SO_2\), \(H_2S\), etc. It occurs at high temperatures. \(O_2\) is the most reactive gas to impart the chemical attack of all the gases mentioned above.

Dry corrosion
(ii). Wet corrosion or Electrochemical corrosion:
The corrosive action in the presence of moisture is called wet corrosion. It happens due to the electrochemical reaction of metal with water or an aqueous solution of salt or acids or bases.

Wet corrosion