
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoHeat is a scalar quantity. The SI unit of heat energy absorbed or evolved is the joule (J).
Other units of Heat energy:
Although the SI unit of heat energy is joule, there are few other units that regularly used . They are,
Although the SI unit of heat energy is joule, there are few other units that regularly used . They are,
Calorie:
One calorie is defined as the amount of heat or thermal energy required to increase the temperature of \(1 gram\) of water through \(1 °C\).
Kilocalorie:
One kilocalorie is defined as the amount of heat or thermal energy required to increase the temperature of \(1 kilogram\) of water through \(1 °C\).
Characteristic features of heat energy transfer:
- Heat always flows from a high-temperature object to a low-temperature object.
- The mass of an object or system is not altered when it is heated or cooled.
- For any heat transfer, the heat gained by the cold system is equal to the heat lost by the hot system.
Heat gained \(=\) heat lost
During the process of heat energy transfer, the object at a lower temperature is heated while the object at a higher temperature is cooled. Thus, sometimes, this process of heat energy transfer is termed as 'Cooling'.
But, in most cases, the term 'heating' is used instead of 'cooling'. When the heat energy is transferred from one object to another, this results in the rise in temperature or lowering of the temperature of either objects.
Heat energy is the agent, which creates the sensation of warmth and makes objects hot. The process in which heat or thermal energy flows from an object at a higher temperature to another object at a lower temperature is known as heating.
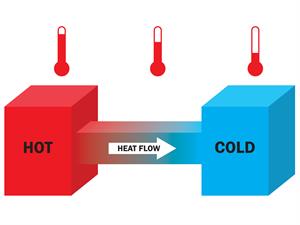
Heat energy transfer from a hot object to a cold object
This process of heat transfer may be done in any of the following ways,
- Conduction
- Convection and
- Radiation
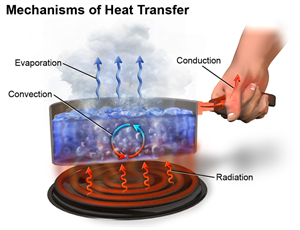
Modes of heat transfer
The above video enables us to understand the measurement of heat.
Reference:
https://commons.wikimedia.org/wiki/File:Heat_Transfer.png