PDF chapter test TRY NOW
Thermal Expansion:
The expansion of a substance on heating is called, the thermal expansion of that substance.
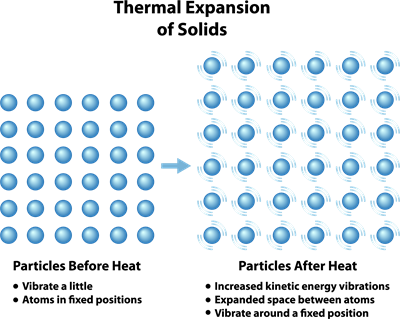
Particles - before and after applying the heat
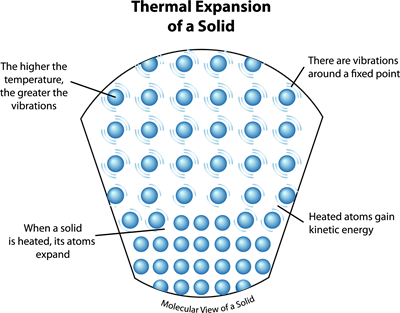
Thermal expansion of a solid
The change in length, area or volume due to contraction or expansion is directly proportional to temperature change.
Example:
We can see an everyday example of thermal expansion in the kitchen.
Almost everyone has had the experience of trying to open a tight metal lid on a glass container, unsuccessfully.
Did you have that experience? What have you done to open it?
Most of you can open it after running hot water over the lid: it gives way and opens at last.

Experiment to understand thermal expansion in solid
Reason:
Answer:
When the hot water is poured over the lid, which is made of steel, having a high coefficient of expansion starts to expand.
On the other hand, glass with a low coefficient of expansion is relatively low expansion compared to the lid. It helps to open the container.
