PDF chapter test TRY NOW
Sublimation occurs when heated, and hence it is called an endothermic process. In an endothermic process, the speed of the molecules is increased, so they move faster. In the endothermic process, the heat is absorbed from the surroundings.
Sublimation:
The process where solid directly changes into vapour on heating is known as sublimation.

Matchstick

Incense sticks
In evaporation, the liquid state changes to a gaseous state.
Sublimation occurs in a substance when the vapour pressure (gas pressure exerted by vapours of the substance) of that substance is high. This method is used for the purification of solids.
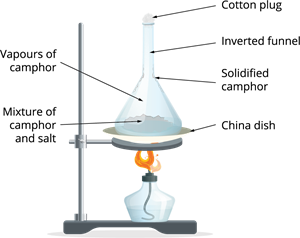
Purification of solids
In the above process, when the substances like camphor and ammonium chloride are heated continuously, forms vapour that is collected in the funnel with the closed end.

Menthol crystals

Naphthalene balls
Certain solid substances like camphor, naphthalene, menthol, and dry ice (solid form of carbon dioxide) that get converted into gas directly upon heating without becoming liquid. This change in the state during the sublimation process is due to the change in temperature of the substances.
Sublimation is a physical change as no new substance is formed. When collected and cooled, the vapours formed will be back to their original state without any change in their chemical properties.
Crystallisation:
When the hot concentrated liquid is cooled, it forms its crystal, and this process is known as crystallisation. In this process, all the soluble impurities are removed.
This is a better technique than evaporation to remove impurities from solids.

Formation of crystals
First, the solute (solid) is dissolved in a solvent and heated continuously, where the excess solvent evaporates. Then, it is allowed to cool down, which forms the crystals. If this process is repeated, it is known as recrystallisation.

Snowflakes
The common examples of the crystallisation process are snowflakes. This process is used in the purification of chemical compounds, seawater etc,.

Sugar Crystals

Cubic crystal of salt
Crystallisation is used to separate and purify substances. In this process, large crystals are formed. The arrangement of particles in the crystal formed is well defined, as in the case of solids. Crystallisation is an example of a physical process.
Reference:
https://upload.wikimedia.org/wikipedia/commons/thumb/5/56/Sugar_2xmacro.jpg/1024px-Sugar_2xmacro.jpg
