PDF chapter test TRY NOW
Combustion
A chemical reaction that takes place in the presence of a fuel and an oxidizing agent is called combustion reaction. The word "combustion" refers to "burning". During combustion reaction, energy is released in the form of heat or light.
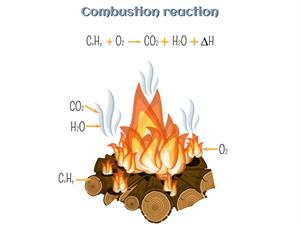
Example for combustion reaction
Combustion reactions are also called exothermic reactions since they release heat energy.
Combustion is one of the first reactions which humans intentionally use.
Oxidation reaction:
Oxidation reaction is a chemical reaction in which oxygen is involved.
In this reaction, the hydrocarbon is combusted with oxygen and typically produces carbon dioxide and water.
Types of combustion:
Combustion is mainly classified into three types:
- Rapid combustion
- Spontaneous combustion
- Explosion
Rapid combustion:
A substance that burns rapidly by external heat and produces heat and light is called rapid combustion. For example, LPG and kerosene stoves.

Examples of rapid combustion
Spontaneous combustion:
A substance that burns without the help of external heat and produces heat and light is called spontaneous combustion. For example, burning of hay, coal when stored in bulk and burning of phosphorous at room temperature.
A substance that burns without the help of external heat and produces heat and light is called spontaneous combustion. For example, burning of hay, coal when stored in bulk and burning of phosphorous at room temperature.

Examples of spontaneous combustion
Explosion:
A substance that burns immediately with the help of pressure or heat producing light and heat with sound is called an explosion. For example, crackers.
A substance that burns immediately with the help of pressure or heat producing light and heat with sound is called an explosion. For example, crackers.

Examples of explosion
Slow combustion:
The combustion that occurs at lower temperature is called slow combustion. For example, respiration.
In the respiration process, food materials are slowly burnt at the low temperatures and produce energy, water and \(CO_2\) like ordinary combustion.
Environmental impacts:
\(CO\) - It causes respiratory problem
\(CO_2\) - It causes global warming
\(SO_2\)/\(NO_2\) - It causes acid rain
