
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoAll living things contain nitrogen that is necessary for their growth. Nitrogen is present in proteins and nucleic acids, the 'building blocks' of all living things.
Swedish chemist Carl Wilhelm Scheele first isolated nitrogen from the air. The name 'nitrogen' is derived from the Greek words 'nitron' and 'gene', meaning 'I produce nitre'. Nitre is a potassium nitrate compound of nitrogen.
Occurrence:
By mass, nitrogen is the fourth most abundant element in the human body and the seventh most abundant element in the universe. Our body is made up of \(3%\) of nitrogen. Titan is the largest moon of Saturn that has an atmosphere made up of \(98%\) nitrogen.
Nitrogen exists in a free and combined state as shown below:
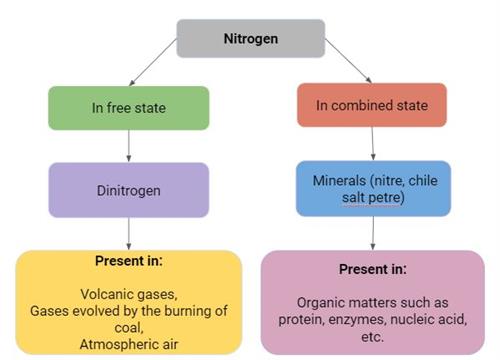
Note: Nitrile is \(KNO_3\) and chile salt petre is \(NaNO_3\)
Physical properties:
- It is a colourless, odourless and tasteless gas.
- Lighter than air.
- Slightly soluble in water.
- It becomes a liquid at a low temperature and looks like water.
- It becomes a white solid when it freezes.
- Neutral to litmus like oxygen.
Uses:
- Nitrogen in the liquid form is used as a refrigerant.
- It gives an inert atmosphere to conduct certain chemical reactions.
- Used for preparing ammonia (by Haber's process), later that is converted into fertilisers and nitric acid.
- Used to fill the space above the mercury in a high-temperature thermometer for reducing the evaporation of mercury.
- It is used in many explosives such as TNT (Trinitrotoluene), nitroglycerin and gun powder.
- Used to preserve fresh foods, manufacture of stainless steel, reducing fire hazards and as part of the gas in incandescent light bulbs.
- Used to inflate vehicle's tyre.
Do you know?
The molecules of nitrogen are larger than normal air molecules; hence, it is harder to leak out from tyres. Therefore, we roll on tires that are always properly inflated, resulting in better fuel economy and longer tire life.

Nitrogen is used to inflate vehicle's tyre