
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoRise in Temperature:
When the substance is heated, molecules in the substance receive heat energy. This heat energy increases the kinetic energy of the molecules. When the molecules receive more energy, the water's temperature increases, confirming that heat energy causes an increase in temperature.
Example:
An activity is explained to understand how heat energy increases the temperature of the object.
Materials Required:
- A small glass bottle
- A rubber cork
- An empty refill
- Water
- Colour
- Candle
- Fork
- Paper
Procedure:
- Take a small glass bottle.
- Fill it with coloured water.
- Make a hole at the centre of the rubber cork.
- Pass empty refill from the hole of the rubber cork.
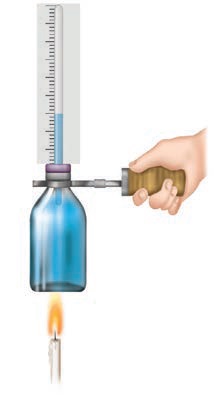
- Make the bottle airtight and notice the water raised in the refill.
- Make a scale on paper, locate it behind the refill, and notice the water's surface position.
- Hold the bottle with a fork, and heat is added to it with the candle. Then observe.
What will happen?
- Initially, the liquid in the refill and bottle are at different temperatures. So, the liquid takes heat energy from the bottle and starts to rise in the refill. This process goes on until they both reach the same temperature (Thermal Equilibrium).
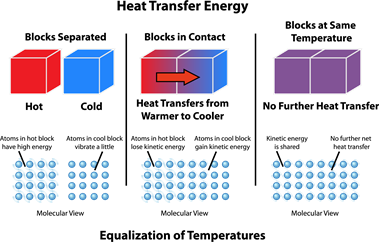
- Now, If you are heating up, it increases the temperature of the bottle, which causes the difference in temperature between liquid and bottle. So, to achieve thermal equilibrium, the heat transfer takes place between the bottle and the liquid. This heat transfer allows the expansion in liquid, which is directly related to a change in temperature. So, we can relate this expansion of the liquid in the refill with the temperature.
- When you cool the bottle, the liquid returns to its initial position due to contraction.