
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoAs we studied earlier, according to Dalton, atoms are indivisible. However, the discovery of electrons and protons (two fundamental particles) within the atom made Dalton's atomic theory meaningless.
It was thought important at the time to understand how electrons and protons are arranged inside an atom. J.J. Thomson was the first one to propose a model of an atomic structure.
J. J. Thomson proposed a model for the atom's structure in order to explain the arrangement of protons and electrons within the atom. This model is known as Thomson's model of an atom.
Thomson stated that an atom's model resembles that of a Christmas pudding.
In a sphere of positive charge, the electrons were like dried fruits in a spherical Christmas pudding.

Thomson's atom model (Christmas pudding)
He also related an atom to a watermelon. The protons are specified by the red component, while the seeds represent the electrons.
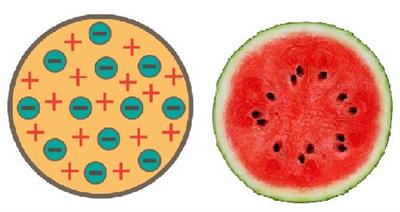
Thomson's atom model (Watermelon)
Thomson proposed:
- The electrons are embedded in a positively charged sphere that represents an atom.
- The magnitudes of the negative and positive charges are the same. Hence, the atom is electrically neutral as a whole.