
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoKazimierz Fajans:
Metal makes an ionic bond with a non-metal, as we all know. Ionic compounds are the result of this process. When the charge of the cation and anion are separated, a chemical is said to be ionic.
In \(1923\), through his X-Ray crystallographic research, Kazimierz Fajans stated that some of the ionic compounds had covalent properties.

Kazimierz Fajans
He developed a series of principles to predict whether a chemical bond is ionic or covalent.
Fajan’s rules are developed by considering the charge of the cation and the relative size of the cation and anion.
- When the cation is small and the anion is large, the bond is more covalent.
- The greater the cation's charge, the greater the covalent character.
Fejans Table:
Ionic | Covalent |
| Low positive charge | High positive charge |
| Large cation | Small cation |
| Small anion | Large anion |
Example:
Sodium chloride has a low positive charge (\(+1\)), a reasonably large cation, and a relatively small anion makes the charges separate. So it is ionic.
In Aluminium triiodide, the higher the positive charge (\(+3\)), the more significant the anion and thus no complete charge separation. So it is covalent.
The relative charge separation of ionic compounds is depicted in the diagram below.
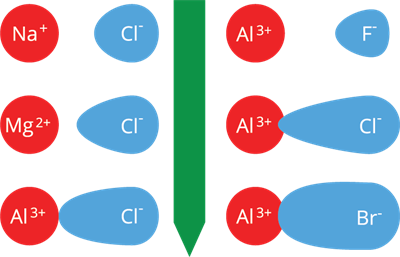
Relative charge separation
Reference:
Fajans image credited by: https://en.wikipedia.org/wiki/Kazimierz_Fajans#/media/File:Kazimierz_Fajans.jpg