PDF chapter test TRY NOW
Coordinate Covalent bond or Dative bond:
In the formation of a normal covalent bond, each of the two bonded atoms contributes one electron to construct the bond.
A covalent bond is formed between two atoms by sharing two electrons, both of which come from combining atoms. This type of bond is known as a Coordinate covalent bond or a Dative bond.
- The lone pair of electrons from an atom in a molecule is usually involved in dative bonding.
- The atom that provides the electron pair is known as the donor atom, while the atom that accepts the electron pair is known as the acceptor atom.
- An arrow () leading from the donor to the acceptor atom represents a coordinate covalent bond.
Formation of coordinate covalent bond:
Consider two atoms, \(A\) and \(B\). Assume that atom \(A\) has an unshared lone pair of electrons and atom \(B\) lacks two electrons in its valence shell.
Atom \(A\) now donates its lone pair, accepted by atom \(B\).
Thus, the lone pair of electrons formerly belonged to atom A is now given by both the atoms, and the bond made by this mutual sharing is called a Coordinate covalent bond \(A\) \(B\).

Coordinate covalent bond formation
Example:
(\(NH_4{^+}\), \(NH_3\) \(BF_3\))
Illustration 1- Formation of a coordinate covalent bond between \(NH_3\) → \(BF_3\) molecules:
- The donated pair of electrons arrive from an already made molecule to another acceptor molecule.
- In this case, the molecule ammonia (\(NH_3\)) contributes a single pair of electrons to the electron-deficient molecule boron trifluoride (\(BF_3\)).
- Thus, \(NH_3\) (donor molecule) and \(BF_3\) (acceptor molecule) form a coordinate covalent bond, which represent \(NH_3\) → \(BF_3\).
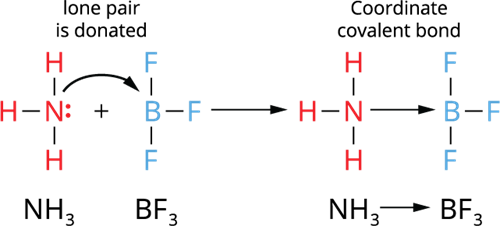
Example of coordinate covalent bond
