
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoDo you know why lemonade tastes the same throughout the drink?
Why and how does this happen?
Whenever we drink lemonade, it tastes the same throughout. It shows that particles of sugar or salt are evenly distributed in the solution (juice).

Lemonade - Solution
Solutions: A solution is a mixture of two or more substances that appears to be uniform in appearance. A solution is a homogeneous mixture of two or more substances.

Solutions
A solution is a mixture in which one substance dissolves fully in the other.
Whereas the solvent is the substance that does not dissolve. Solvent is a variable that is present in a greater quantity by weight.
And, the solute is the material that dissolves. A solute is a part of a solution that is present in a smaller quantity by weight.
If we add the solvent and solute, we get the solution.
Solution \(=\) Solvent \(+\) Solute.
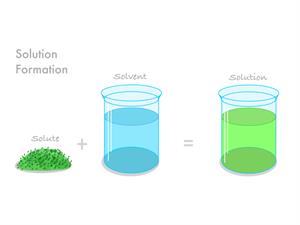
Solution formation
Types of solutions:There are various types of solutions based on their medium of dispersion.
(a) Solid in a solid solution: Alloys
(b) Solids in a liquid solution: Salt solution, sugar solution
(c) Liquids in a liquid solution: Lemon extract in water
(d) Gas in a gas solution: Natural gas, air
(e) Gas in a liquid solution: Carbonated drinks
Example:
1. Solids in a liquid solution: Salt solution, sugar solution.

Salt solution
Solute: Salt is solute.
Solvent: Water is solvent.
2. Gas in a liquid solution: In aerated drinks.

Carbonated drinks
Solute: Carbon dioxide (gas) is solute.
Solvent: Water (liquid) is solvent.
Solvent: Water (liquid) is solvent.
3. Solid in a solid solution: Alloys.
Alloys are mixtures of two or more metals, a metal and a non-metal, that cannot be segregated into their constituent parts using physical methods.

Objects made from Brass
However, since an alloy exhibits its constituents' properties and may have a variable composition, it is still called a mixture. For example, brass is made up of \(30\%\) zinc and \(70\%\) copper.
4. Liquids in a liquid solution: In lemonade

Lemonade
Solute: Lemon extract is solute.
Solvent: Water is solvent.
Solvent: Water is solvent.
5. Gas in a gas solution: Natural gas, air etc.
Air is a gaseous mixture in a gas solution. It is homogeneous in composition.

Wind
Oxygen \(21\%\) and nitrogen \(78\%\) are the two primary constituents of air. The other gases are only found in trace amounts \(11\%\). Natural gas is also an example of this type of gas in a gas solution.