
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoLimitations of the Mendeleev table:
- Elements with significant differences in properties were grouped. (Soft metals like sodium and potassium were mixed in with hard metals like copper and silver).
- The element hydrogen could not be assigned to a specific position. (Hydrogen was placed in between the metals.)
- Throughout the experiment, the increasing order of atomic mass was not strictly followed.
- Isotopes have no place in the periodic table.
Germanium Properties:
Property | Mendeleev’s prediction (\(1871\)) | Actual property (\(1886\)) |
| Atomic Mass | About \(72\) | \(72.59\) |
Specific Gravity | \(5.5\) | \(5.47\) |
| Colour | Dark grey | Dark grey |
| Formula of Oxide | \(EsO_2\) | \(GeO_2\) |
| Nature of Chloride | \(EsCl_4\) | \(GeCl_4\) |
Modern periodic table:
In \(1913\), the English physicist Henry Moseley proved that the properties of elements are determined by their atomic number, not by atomic mass. As a result, the modern periodic table was created by arranging elements in increasing atomic number order. This modern periodic table extends Mendeleev's original periodic table, known as the extended periodic table.
Modern periodic law:
An element's atomic number (Z) represents the number of protons (positive charge) or electrons (negative charge).
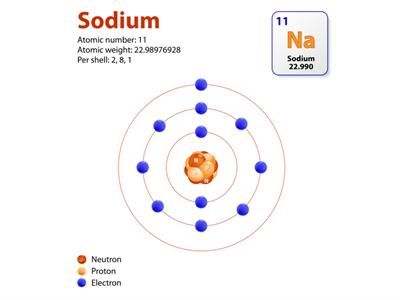
Elements' physical and chemical properties are determined by the number of protons and electrons and how they are arranged in atoms.
According to modern periodic law, the chemical and physical properties of the elements are the periodic functions of their atomic numbers.
Characteristics of the modern periodic table:
- All the elements are organized in the increasing order of their atomic number.
- The horizontal rows are referred to as periods. The periodic table contains seven periods.
- Periods are assigned to the elements depending on the number of shells in their atoms.
- Groups are vertical columns in the periodic table that run from top to bottom.
- Based on their properties of elements, they are grouped into various families.
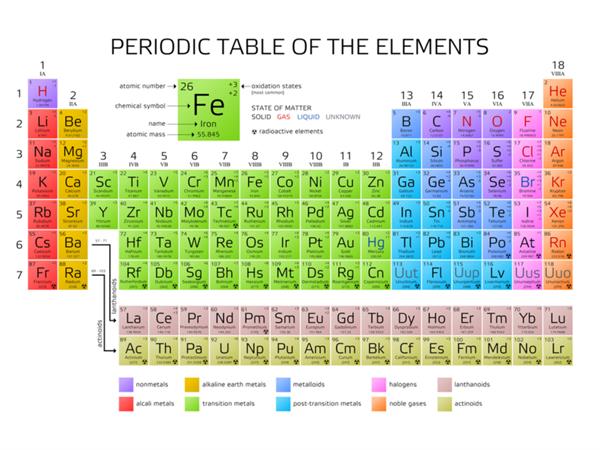
Groups in modern periodic table:
| Group | Families |
| \(1\) | Alkali metals |
| \(2\) | Alkaline earth metals |
| \(3\) to \(12\) | Transition metals |
| \(13\) | Boron Family |
| \(14\) | Carbon Family |
| \(15\) | Nitrogen Family |
| \(16\) | Oxygen (or) Chalcogen Family |
| \(17\) | Halogens |
| \(18\) | Noble gases |