PDF chapter test TRY NOW
In addition to carbon, organic compounds contain atoms such as hydrogen, oxygen, nitrogen, and so on that bond with carbon. When these atoms are combined with carbon, they form various classes of organic compounds.
Let us now look at various classes of organic compounds:
Hydrocarbons:
Hydrocarbons are organic compounds that are made up of only carbon and hydrogen atoms. The carbon atoms combine to form the compounds' framework. These are considered the parent organic compounds, and all other compounds are thought to be derived from them by replacing one or more hydrogen atoms with other atoms or groups of atoms.
Hydrocarbons are further classified into three classes:
- Alkanes
- Alkenes
- Alkynes
Alkanes:
Alkanes are hydrocarbons with only a single bond. Alkanes are saturated compounds because they all have single bonds (\(C-C\)).
Alkanes are represented by the general formula \(C_nH_{2n + 2}\) (where \(n = 1, 2, 3,\) and so on).
Methane (\(CH_4\)) is the simplest alkane (for \(n=1\)).

Alkanes
Alkenes:
Alkenes are hydrocarbons that contain one or more carbon to carbon bonds (\(C=C\)). Alkenes are unsaturated compounds because they have double bonds.
The general formula (\(C_nH_{2n}\)) is used to represent them.
The simplest alkene is ethylene (\(C_2H_4\)), which has two carbon atoms (\(n=2\)).

Alkenes
Alkynes:
Alkynes are hydrocarbons with carbon to carbon triple bonds (\(C\equiv C\)). Alkynes are unsaturated due to the presence of a triple bond between carbon atoms.
Alkynes are represented by the general formula (\(C_nH_{2n – 2}\)).
Acetylene (\(C_2H_2\)) is the simplest alkyne, with only two carbon atoms.
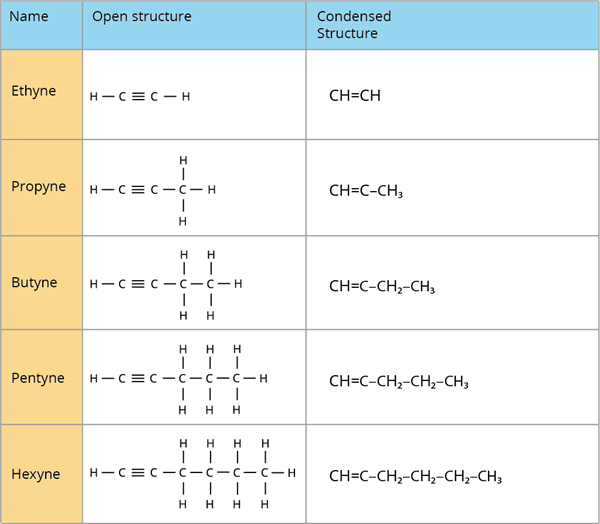
Alkynes
Hydrocarbons with \(1\) to \(5\) carbon atoms:
Number of carbon atoms | Alkane \(C_nH_{2n + 2}\) | Alkene (\(C_nH_{2n}\)) | Alkyne (\(C_nH_{2n – 2}\)). |
\(1\) | Methane (\(CH_4\)) | - | - |
\(2\) | Ethane (\(C_2H_6\)) | Ethene (\(C_2H_4\)) | Ethyne (\(C_2H_2\)) |
\(3\) | Propane (\(C_3H_8\)) | Propene (\(C_3H_6\)) | Propyne (\(C_3H_4\)) |
\(4\) | Butane (\(C_4H_{10}\)) | Butene (\(C_4H_8\)) | Butyne (\(C_4H_6\)) |
\(5\) | Pentane (\(C_5H_{12}\)) | Pentene (\(C_5H_{10}\)) | Pentyne (\(C_5H_8\)) |
Note: Methene and Methyne do not exist because they have only one carbon atom. These compounds cannot form multiple bonds, as hydrogen can share only one electron.
