
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoThere are millions of known organic compounds, and many new organic compounds are discovered in nature or synthesised in the laboratory every year. This may greatly obscure organic chemistry.
However, each compound can be assigned a unique molecular structure, which can then be listed using systematic classification methods and eventually named based on its structural arrangements.
Chemists recognised early on that compounds with similar structural features have identical chemical properties. As a result, they began to classify compounds based on their common structural arrangements.
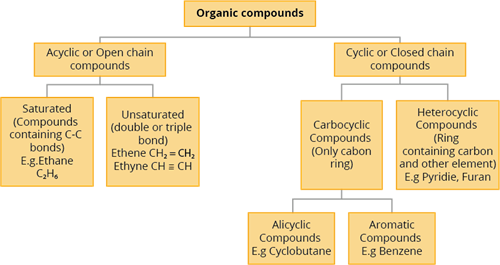
Classification of organic compounds
Organic chemistry is the study of carbon compounds that have been catenated. Organic compounds contain carbon atoms that are linked together by covalent bonds and thus exist as chains.
Organic compounds are classified into two types based on the skeleton of the carbon chain:
- Acyclic or Open-chain compounds
- Cyclic or Closed-chain compounds
1. Acyclic or Open-chain compounds:
The compounds in which the carbon atoms form a chain-linked in a linear pattern or branched are called as acyclic or open-chain compounds.

Open-chain compounds
Open-chain compounds are further classified as:
a) Saturated compounds: The compound is saturated if single bonds connect all of the carbon atoms in the chain. In this, all the valencies of carbon are satisfied by single bonds.
b) Unsaturated compounds: If there are double or triple bonds between the carbon atoms, the compound is unsaturated. In this, all the valencies of carbon are satisfied by multiple bonds.

Open-chain compounds
2. Cyclic or Closed-chain compounds:
Cyclic compounds are organic compounds in which the carbon atom chain is closed or cyclic.
In heterocyclic compounds, the chain contains carbon as well as other atoms such as oxygen, nitrogen, sulphur, and so on.
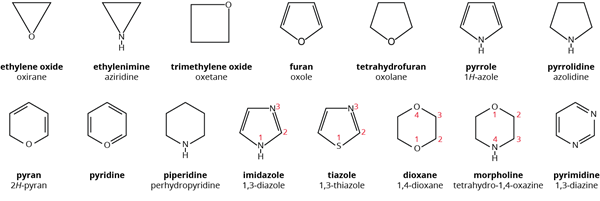
Heterocyclic compounds
Carbocyclic (Homocyclic) compounds are those in which the chain contains only carbon atoms.
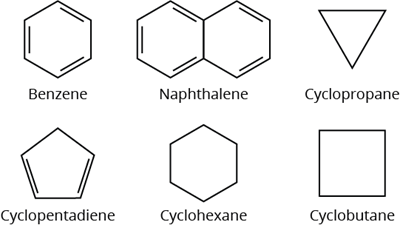
Homocyclic compounds
Carbocyclic compounds are further classified as:
- Alicyclic compounds
- Aromatic compounds
Alicyclic compounds have one or more carbocyclic rings that can be either saturated or unsaturated.
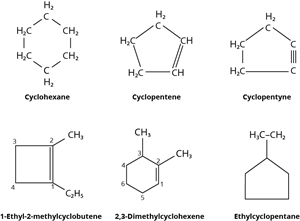
Alicyclic compounds
Aromatic compounds have one or more benzene rings (rings containing alternate double bonds between carbon atoms).
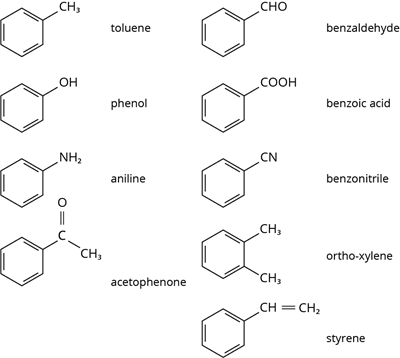
Aromatic compounds