
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoThe pressure difference can also happen if there is any change in the temperature.A balloon is taken and tightly stretched over the neck of the boiling tube. It is then inserted into the beaker containing hot water.
Air expands on heating:

A balloon covering the neck of a tube
After \(2\) \(–\ 3\) minutes, a change in shape of the balloon is observed. The balloon is inflated since the hot air present inside the tube heats the air around it. This heating expands the air and eventually inflates the balloon by filling the space inside.

Inflation of the balloon
Generally, gases or air expand more than solid and liquid on heating. The gases that expand on heating occupy more space and thus, become lighter.
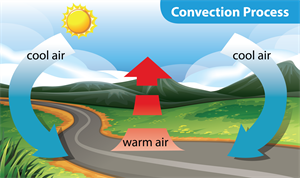
Movement of warm and cold air
The warm air is always lighter than the cold air. When the warm air rises from a particular place, a lower air pressure is created at that place. Eventually, the colder air that surrounds the area rushes into that space and fills it up.
\(\)
\(\)
Convection: 
Convection is a mode of heat transfer in which the warm air rises, and the air from the sides moves in to fill the space.
Example:
Smoke rises in the sky due to convection.

Smoke from the chimneys
Air contracts on cooling:
Now, the boiling tube is taken out of the beaker and allowed to cool for some time. This time it is inserted in another beaker containing ice-cold water. After \(2\) \(–\ 3\) minutes again, there is a change in the shape of the balloon. It is observed that the balloon is deflated since the air present inside the tube contracts on cooling.

Deflation of the balloon
Hot air experiment:
Two paper bags or cups of the same size are taken and hanged on a wooden or metal stick. The paper bags (cups) are placed in the inverted position on the ends of a stick. A piece of thread is tied in the middle of the stick to hold it like a balance.
Two paper bags or cups of the same size are taken and hanged on a wooden or metal stick. The paper bags (cups) are placed in the inverted position on the ends of a stick. A piece of thread is tied in the middle of the stick to hold it like a balance.
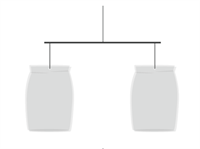
Experimental setup
A burning candle is kept under one of the bags (cups). It is noticed that the paper bag (cup) placed above the candle is pushed upwards. This is because the warm air being lighter rises and fills the space inside the bag.
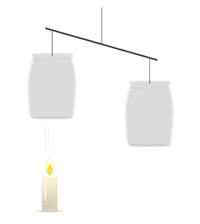
Warm air pushes the paper bag upwards
Therefore, one side of the stick becomes lighter, moves upwards and thus, the balance is disturbed. From this experiment, it is evident that warm air rises. This concept is applied in the launching of hot air balloons.

Hot air balloons