
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoHistory of Candle:
 |
Candle flame
It is said that the Ancient Romans invented wicked candles. In \(3000\) B.C, Egyptians used wicked candles made from papyrus rolled and dipped in melted beeswax or tallow.
Likewise, in early civilisation, these wicked candles were made from plants and animals. In China, wax is made from the indigenous insect with seeds mixed, and the wick is made from rolled rice paper.
In Japan, the wax is extracted from tree nuts, whereas in India, the resin is made from cinnamon trees boiling their fruits.
Michel-Eugene Chevreul, a French chemist, produced candles from stearic acid, which was separated from fatty acid of glycerin in the \(19th\) century.
In modern times, candles are made easily with different shapes and sizes and even cleaner than times by using paraffin wax as the byproduct of petroleum in the refining process.
Characteristics of Wax:
- Made of hydrocarbons (a byproduct of petroleum)
- Low melting point
- Insoluble in water
- Solid at room temperature (when it is ignited, it is in liquid form)
Flame:
 |
When a substance is on fire, the stream of hot burning gas or the substance which vaporises on burning is called as flame. It is the visible part of the fire, and there are different zones of flame based on temperature and hazard.
The wax reacts with atmospheric oxygen and forms carbon dioxide and water (in the form of steam) with some smoke (due to unburnt carbon mixed with steam produced).
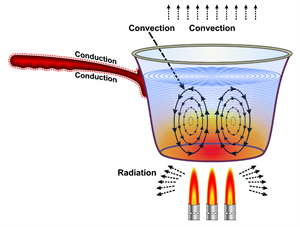 |
Heat transfer forms
Here the heat is transferred in all three forms such as conduction, convection, and radiation. The heat from the wick is carried to the wax, and the wax melts. It is known as conduction.
The vapours from the wick heat the air surrounding the candle, and cooler air replaces, and the process continues known as convection, and the emission of heat energy in the form of rays or waves is known as radiation. The chemical reaction that takes place is combustion.
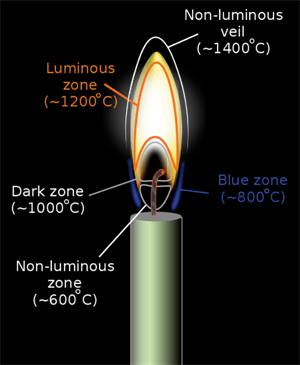
Zones of Flame:
There are different zones of flames based on the temperature and hazards. They are
- Inner Zone (Dark flame)
- Middle Zone (Luminous flame)
- Outer Zone (Non-luminous flame)
 |
Flame zones
Inner Zone (Dark):
In this zone is around the wick, there is no combustion and comparatively the least hot site (as there is unburnt carbon mixed with steam forms of aerosol which causes pollution) and it is dark or black in colour.
Middle Zone (Luminous):
In this zone, partial combustion takes place, moderately hot, the flame is yellow in colour and is the significant portion of the flame. In this zone, there is an insufficient amount of oxygen present.
Outer Zone (Non-luminous):
In this zone, complete combustion takes place, the hottest region of the flame and the colour of the flame is blue. This is the non-luminous zone (do not emit light) of the flame. In this zone, there is a sufficient amount of oxygen present.
Note: All three essential elements are required for burning, and the absence of even one element ceases the burning or combustion process. The wax is converted to heat, light and chemical waste.
Reference:
A loose necktie, CC BY-SA 4.0 <https://creativecommons.org/licenses/by-sa/4.0>, via Wikimedia Commons