
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoLet us try to name some of the linear and substituted hydrocarbons in a systematic manner using IUPAC rules:
1. \(CH_3-CH_2-CH_2-CH_2-CH_3\)
Step 1: According to rule \(1\), there is a five-membered carbon chain; hence, the root word is ‘Pent’.
Step 2: According to rule \(2\), all the bonds between carbon atoms are single bonds, and thus the suffix is ‘ane’.
So, the name of the given compound is \(\text{Pent + ane = Pentane}\).
2. 

Step 1: According to rule \(1\), there is a five-membered carbon chain; hence, the root word is ‘Pent’.
Step 2: According to rule \(2\), there is a substituent. So, the carbon chain is numbered from the left end, which is closest to the substituent.

Step 3: All are single bonds between the carbon atoms, and thus the suffix is ‘ane’.
Step 4: According to rule \(6\), the substituent is a methyl group compound, located at the second carbon atom.
So, its locant number is \(2\). Thus the prefix is ‘\(2-Methyl\)’.
Hence, the name of the given compound is \(\text{2-Methyl + pent + ane = 2-Methyl pentane}\).
3. 

Step 1: According to rule \(1\), there is a seven-membered carbon chain; hence, the root word is ‘Hept’. Step 2: According to rule \(2\), there is a substituent. So, the carbon chain is numbered from the left end, which is closest to the substituent.
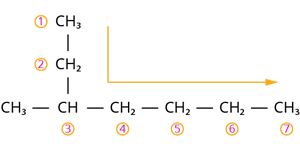
The correct way of numbering the carbon atoms

The wrong way of number the carbon atoms
Step 3: All are single bonds between the carbon atoms, and thus the suffix is ‘ane’.
Step 4: According to rule \(6\), the substituent is a methyl group compound located at the third carbon atom.
So, its locant number is \(3\). Thus the prefix is ‘\(3-Methyl\)’.
Hence, the name of the given compound is \(\text{3-Methyl + hept + ane = 3-Methyl heptane}\).
4. 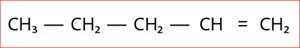
Step 1: According to rule \(1\), there is a five-membered carbon chain; hence, the root word is ‘Pent’.
Step 2: According to rule \(2\), there is a carbon to carbon double bond (\(C=C\)), and thus the suffix is ‘ene’.
Step 3: According to rule \(3\), the carbon chain is numbered from the end of the double bond that has the lowest locant number, as shown below
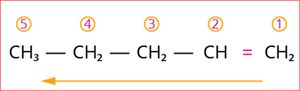
Numbering starts from the double bond.
Step 4: The locant number is \(1\) for the double bond, and hence the suffix is ‘\(-1-ene\)’.
Hence, the name of the given compound is \(\text{Pent + (-1-ene) = Pent-1-ene}\).
These are some of the examples for naming the hydrocarbons according to IUPAC rule. In the next section, we will see the IUPAC naming for various other compounds.