PDF chapter test TRY NOW
From olden times, mankind was enthralled by the vast universe and its constituents. Likewise, the human mind has been pursuing the smallest particle perpetually and has discovered a vast new world of sub-atomic particles.

The vast universe and an atom
As a result of all the experiments and research, a whole new area of physics emerged, leading to revolutionary discoveries and inventions such as the radio, transistors, X-rays, computers, etc. This new area of physics was referred to as Modern physics.
Nuclear physics is one such development of modern physics.
Nuclear physics is the study of atomic nuclei and their constituents (protons and neutrons).
Discovery of elementary particles:
Humans have always had a desire to learn about atoms. Everything in the universe is made up of atoms.
In \(400\ BC\), a Greek philosopher named 'Democritus' said that matter comprises small indestructible units known as atoms.
In \(1803\), John Dalton proposed that elements are made up of similar atoms. Later Goldstein discovered positive rays, which was named by Rutherford as protons. In the meanwhile, J J Thomson discovered cathode rays, also known as electrons.
In \(1932\), the chargeless particles known as neutrons were discovered by James Chadwick. A huge number of elementary particles, such as the photon, meson, positron, and neutrino, have been identified recently.
In \(1911\), a British scientist named Ernest Rutherford proposed that the atom's mass is concentrated in its central part, known as the nucleus.
Atom:
Atoms are mainly composed of particles such as electrons, protons, and neutrons. In an atom, the negatively charged particles are the electrons, the positively charged particles are the protons, and the chargeless particles are the neutrons.
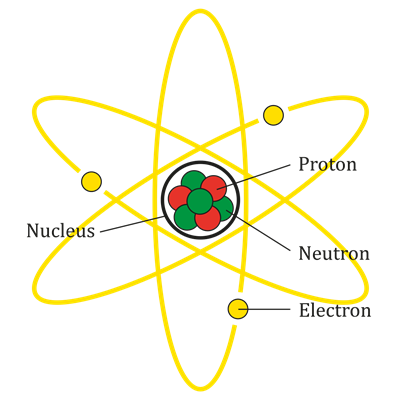
An atom
An atom consists of a nucleus with positively charged protons and chargeless neutrons. Also, the negatively charged electrons orbit the nucleus.
