PDF chapter test TRY NOW
In the disintegration process, the nucleus that disintegrates is referred to as the parent nucleus, and the nucleus generated after the disintegration is referred to as the daughter nucleus.
\(\)
\(\)
The particle that induces the artificial disintegration is known as the projectile, and the particle produced after the disintegration is known as the ejected particle.
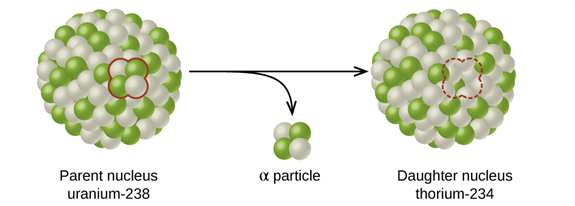
The disintegration of uranium to thorium with an alpha particle
When hit by a projectile, the parent nucleus gets converted into an unstable nucleus and decays spontaneously. During the decay, the unstable nucleus emits the daughter nucleus along with an ejected particle.
If \(X\) is represented as parent nuclei and \(Y\) is represented as daughter nuclei, then the nuclear disintegration is denoted as \(X\ (P, E)\ Y\). Here, \(P\) denotes the projectile particle, and \(E\) denotes the ejected particle, respectively.
Example:
In the given nuclear reaction, is unstable and radioactive in nature. Hence, the reaction can be denoted as , where \(\alpha\) is the projectile particle, and \(n\) is the ejected particle, respectively.
The un-stability in the nucleus of heavy elements leads to spontaneous radioactive disintegrations.
Artificial transmutation:
Artificial transmutation is the process of transformation of one element into another element by artificial disintegration means of colliding it with high energy particles.
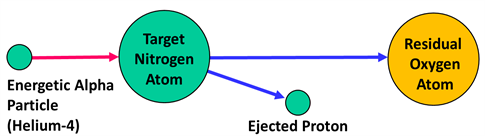
Artificial transmutation of nitrogen
Such type of artificial transmutations can be achieved by using α-particles, protons, neutrons or even smaller nuclei as the bombarding or colliding particles.
Artificial transmutation by alpha particles:
Artificial transmutation of fluorine and aluminium is possible by using the \(α\)-particles as projectiles.
Artificial transmutation of fluorine and aluminium is possible by using the \(α\)-particles as projectiles.
Artificial transmutation by protons:
A nucleus of a hydrogen atom or proton is used as a projectile to transmute lighter elements such as lithium and sodium.
A nucleus of a hydrogen atom or proton is used as a projectile to transmute lighter elements such as lithium and sodium.
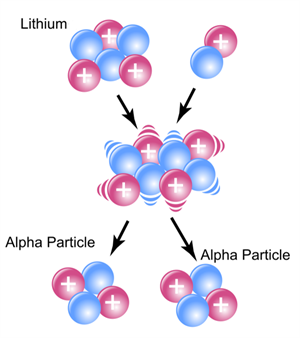
Artificial transmutation of lithium
Reference:
https://upload.wikimedia.org/wikipedia/commons/1/1a/CNX_Chem_21_03_Reaction1.png
https://upload.wikimedia.org/wikipedia/commons/c/ce/Patrick_Blackett_1925_Experiment_Interpretation_as_Drawn_by_Steven_B._Krivit.png
