PDF chapter test TRY NOW
Properties of Alpha, Beta and Gamma rays:
Properties | Alpha rays | Beta rays | Gamma rays |
What are they? | Helium nucleus (\(_{2}He^{4}\)) with two protons and two neutrons | Electrons (\(_{-1}e^{0}\)) | Electromagnetic waves consisting of photons |
Charge | Positively charged particles with charge of \(+2e\) | Negatively charged
particles
with charge of \(–e\) | Chargeless neutral particles |
Ionising power | \(100\) times greater than \(β\) rays
and\(10,000\) times greater
than \(γ\) rays | Comparatively low ionisation power | Very less ionisation power |
Penetrating power | Low penetrating power | Penetrating power is greater than that of \(α\) rays | Very high penetrating power greater than that of \(β\) rays |
Effect of electric and magnetic field | Deflected by both the fields | Deflected by both the
fields but opposite
to that of alpha rays | Not deflected |
Speed | Ranges from \(1/10\) to \(1/20\) times the speed of light | Has \(9/10\) times the speed of light | Travel with the speed of light |
| Rest mass | \(4\) times the mass of the proton | Equal to the mass of an electron | No mass |
| Stopping substance | Thin paper and human skin | About \(1\ mm\) of lead or \(3\ mm\) of aluminium | About \(30\ cm\) of iron or few metres of concrete |
| Biological damage | Causes damage and harmful to humans | Causes damage and harmful to humans | Passes through a human body and destroy the cells (kill cancer cells) |
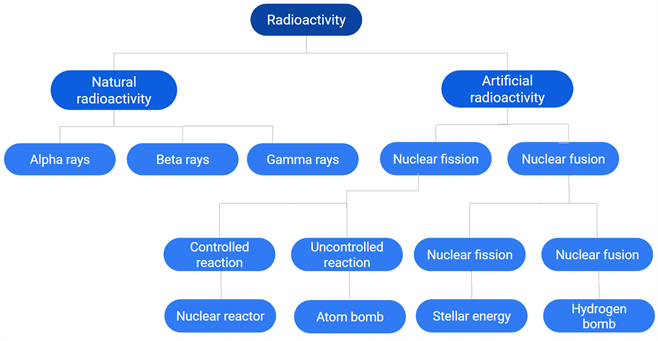
Mind map of Nuclear Physics: