
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoAtomic radius:
The atomic radius is defined as the distance between the nucleus centre and the outermost shell containing the valence electron.
The radius of a separated atom is not possible to measure directly.

Except for noble gases, the atomic radius is usually referred to as a covalent radius or metallic radius depending on the bonding between the concerning atoms.
Metallic radius:
The atomic radius in metal atoms is known as metallic radius. It's defined as half the distance between adjacent metal atoms' nuclei.

Figure: (a). Metallic Radius (b). Covalent radius
Covalent radius:
Covalent radius is a term used to describe the atomic radius of nonmetallic elements. It is defined as half the distance between the adjacent nuclei of two covalently bonded atoms of the same element in a molecule.
For example, let us consider the \(H_2\) molecule. The distance between the two hydrogen nuclei of the molecule is \(0.74\) Å. So its covalent radius is \(0.74/2\) = \(0.37\) Å. When you see the variation of the atomic radii in the periodic table, there are two different trends.
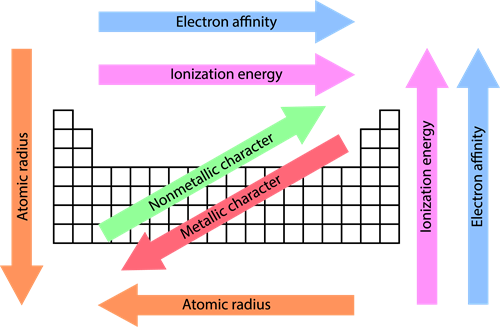
From left to right, the atomic radius of the elements decreases, whereas along with the groups, from top to bottom, the atomic radius increases.
The positive charges impose a strong attraction over the electrons, and thus the electron cloud shrinks towards the nucleus, which results in a decrease in the atomic size.
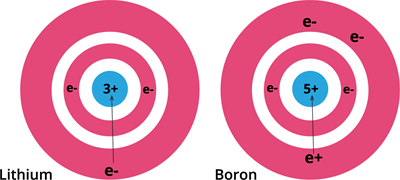
Variation of atomic radius.
The above figure shows how the atomic radius decreases from lithium to boron.
Ionic radius or radii:
It is defined as the distance from the centre of the nucleus of the ion up to the point where it exerts its influence on the electron cloud of the ion.

- You know that ions are formed when an atom lose or gain electrons. For example, when a neutral atom loses an electron, it becomes a positively charged ion called a cation. In contrast, the gain of an electron by a neutral atom forms a negatively charged ion called an anion.
- The size of the ions is essential to determine their behaviours in solutions and the structure of ionic solids.
- The size of a cation is always smaller than its corresponding neutral atom. But, the anion is more significant than its neutral atom.
Important!
Note: The cation's size decreases as the positive charge increases. The size of the anion increases as the negative charge increases.
For example, lithium and sodium lose a single electron from their outermost energy level to form cations. The ions formed are smaller because the remaining electrons are at inner cells and attracted more strongly by the nucleus.
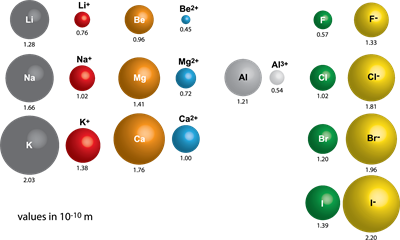
Fluorine and chlorine form negative ions by gaining an electron. When electrons are added, the charge on the nucleus is insufficient to hold the extra electrons as tightly as it holds the electrons in the neutral atom.
So, as seen in atomic radius, ionic radii also decrease from left to right and increase down the group.