
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoConcentrated and dilute solutions is a different type of classification of unsaturated solutions. It expresses the relative concentration of two solutions in terms of the solutes present in a given amount of solvent.
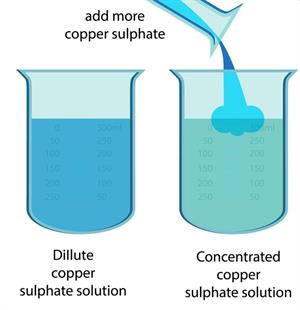
Dilute and concentrated solution
For instance, you are given two cups of tea. When you taste them, one seems to be sweeter than the other.
What conclusion do you draw from it?
The sugar content of the sweeter tea is higher than that of the other.
What words can you use to express your observation?
You could say that the 'tea is stronger'.
A chemist, on the other hand, would say the tea is ‘concentrated.’
When two solutions with the same solute and solvent are compared, the one with a higher amount of solute per given amount of solvent is referred to as a ‘Concentrated solution.’ In contrast, the other is referred to as a ‘Dilute solution.’
The distinction between dilute and concentrated solutions is a qualitative representation. It does not imply the amount of solute. This distinction is visible through physical characteristics such as colour, density, and so on.
Look at the following pictures. Label them as dilute and concentrated solutions,and justify your answer:

The dark coloured solution of tea and copper sulphate is the concentrated, while the lighter coloured one is the diluted solution.

Concentrated solution

Dilute solution
In the above image, the solution marked as ‘(a)’ in both tea and copper sulphate is the concentrated solution, whereas ‘(b)’ is the dilute solution.
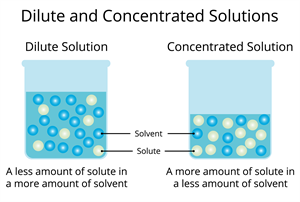
Types of solution based on solute
When the solute is higher than the solvent, we can say it is a concentrated solution. In contrast, if the solvent is higher than the solute, it is known as a dilute solution.
Reference:
https://stocksnap.io/photo/tea-teabags-NYSS2K287L
https://www.pxfuel.com/en/free-photo-eqkhd