
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoPhysical change: In the physical transformation, only the physical properties such as colour, structure, size are changed. No new substances are formed. They are reversible changes.
Example:
Freezing, melting, evaporation etc.
Chemical change: Any substance that changes its chemical properties is known as chemical change. In this change, a new substance is formed, and these changes are irreversible in nature. These changes are permanent.
Example:
Photosynthesis reaction, burning of a matchstick, paper, etc.
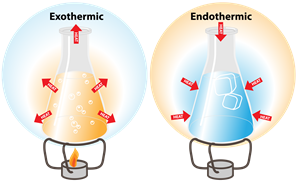
Exothermic and endothermic reaction
1. Exothermic change:
- The heat is released in this reaction.
- Heat is the product.
Combustion, condensation, freezing, neutralisation reaction, dissolving an acid in water are best examples of exothermic changes as heat is released for the change to occur.
This is due to the bond formation, which causes to release of heat. The formation of bonds requires less energy.
Combustion:

Release of heat
The burning of wood and paper releases heat energy to their surroundings, which is the best example of exothermic chemical change. The product formed is different from the reactants. This is an irreversible change.
Freezing:

Water kept in a freezer
The water molecules release heat to their surroundings and freezes to form ice. This is an example of exothermic physical change as this process can be reversed.
2. Endothermic change:
- The heat is absorbed in this reaction.
- Heat is the reactant.
Cooking, photosynthesis, evaporation are the best examples of endothermic changes as heat is absorbed for the change to occur.
The breaking of the bond requires more energy which causes heat to be absorbed.
Cooking:

Absorption of heat
Making dosa from batter is the best example as heat is absorbed from the pan, making the batter turn into the dosa. Similarly, making vada is also an example of endothermic chemical changes.
Evaporation:

Heat is absorbed, and water evaporates as vapour
The water molecule absorbs heat and evaporates as vapours. This is an example of endothermic physical change as the process is reversible.
What is enthalpy?
Enthalpy: The change in heat energy during the reaction is known as enthalpy. The value of enthalpy differs with heat.
In case if heat is absorbed, the enthalpy is positive and negative if heat is released.
Exothermic reaction:
Endothermic reaction:
Exothermic and endothermic reaction