
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoIons:
When an atom or a group of atoms lose or gain electrons, they are converted into ions or radicals.
The two types of ions are cations (+) and anions (-).
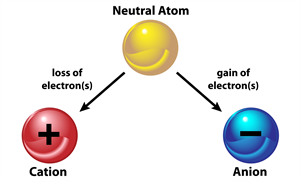
Types of ions
- The number of protons in an atom equals the number of electrons, giving it electrically neutral.
- However, atoms attempt to achieve a stable electronic configuration (duplet or octet) during chemical reactions by obtaining or losing one or more electrons based on valency.
- When an atom gains an electron, it increases the electron count and hence has a negative charge.
- When an atom loses an electron, it gains a more significant number of protons, resulting in a positive charge.
- These atoms which give positive or negative charges are called ions.
- When an atom loses an electron, we assign the ‘+’ sign to it.
- When an atom gains an electron, we assign the ‘-’ sign to it.
Ions