
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoThe gold-leaf electroscope was developed by a British scientist named Abraham Bennet in the year \(1787\). Gold and silver are preferred in electroscopes because they are the most excellent conductors of electric current. The electroscope is used to detect the presence of charges on a body.
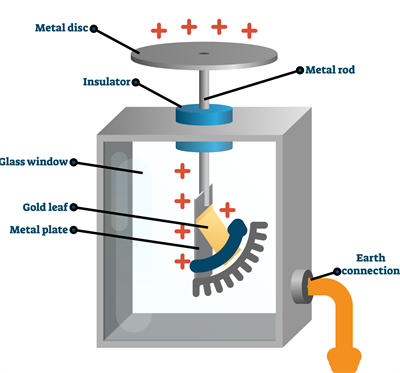
Structure of an electroscope:
- The electroscope is made up of a glass jar or a glass box.
- A vertical brass rod is inserted into the glass jar through a cork (insulator).
- The head of the brass rod has a horizontal brass rod or a brass disc.
- Two gold leaves are hung from the brass rod inside the jar.

Sample model of a gold-leaf electroscope
Working of an electroscope:
- When a charged object touches the electroscope's brass disc, the electric charge gets transferred to the gold leaves through the rod. This results in the gold leaves moving away from each other as like charges repel each other.
- The movement of the leaves shows the presence of charges in a body.
Shows the working of the gold leaves when a charged body comes into contact
Charging:
- The transfer of charges from one object to another is called charging.
- In the case of the gold leaves, the charge is transferred through the brass rods.
Electrical discharge:
- The gold leaves return to their normal position after some time. This happens because they lose their charge after a while. This process is called electrical discharge.
- The charges in the gold leaves would also be discharged when someone touches the brass rod with empty hands. In that case, the charges are transported to the earth through the human body.