
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoIn conduction, heat transfer takes place between two ends of the same solid or between two solid substances that are at different temperatures but in contact with each other. Thus, we can define conduction as the heat transfer in solids from the higher temperature region to the lower temperature region without any actual movement of atoms or molecules.
Particles with higher kinetic energy transfers thermal energy towards the lower kinetic energy particles. High-speed particles clash with particles moving at a slow speed. As a result, slow speed particles increase their kinetic energy. This is a typical form of heat transfer and takes place through physical contact.
Heat conduction is a process in which heat is transferred from the hotter part to the colder part of a body without involving any actual movement of the molecules of the body.
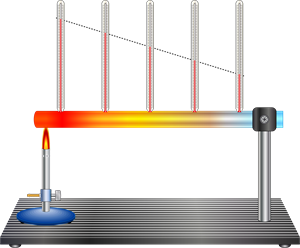
Example:
The following are some of the examples of conduction in daily life.
- In our homes, we cook food in vessels made up of metals. When vessels are heated, heat is transferred from the metal to the food.
- In our homes, when we iron dresses, heat is transferred from iron boxes to clothes.
Based on the substance's ability to conduct heat, substances are classified as:
- Conductors
- Insulators
Conductors:
Substances that conduct heat easily are called conductors. All metals are good conductors of heat.
Insulators:
Substances that do not conduct heat are called insulators. Wood, cork, cotton, wool, glass, rubber, etc., are insulators.
Example:

- Cooking Vessels are made up of metals because they are good conductors of heat.
- Handles of cooking vessels are made up of plastic or wood because they are poor conductors of heat.