
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoAll scientific and technological experiments begin with measurement. The quality of a measurement can be described using terms such as
- Error
- Accuracy
- Precision
- Approximation and
- Roundoff
Error
Every measurement has some value of uncertainty. This uncertainty is referred to as an error. The difference between the real value and the observed value is known as error.
Accuracy
Accuracy is the closeness of a measured value to the actual value or true value. It is usually needed while taking measurements.
Illustration
Imagine you purchased \(3.5\ kg\) of apple in a shop by measuring it in a traditional beam balance. On the way to your home, you weigh the purchased apples again in a different shop using a digital balance. Now, the apples weigh only \(3.3\ kg\).

Here, this measurement is considered inaccurate. But, if the purchased apple measures the same \(3.5\ kg\), then the measurement is considered as an accurate one.
Precision
Precision is the closeness of two or more measurements to each other.
Illustration
Imagine the pack of apples is weighed five or more times. If the apples weigh \(3.5\ kg\) each time, then the measurement is very precise. If the measurement varies, then it is considered imprecise.
Important!
Precision and accuracy may appear to be the same thing. However, the two terms are not the same.
Illustration
Take a look at the arrows fired by three people.
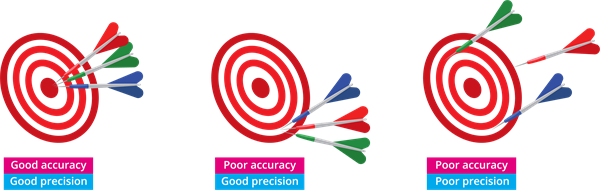
- In the first image, all of the arrows hit in the centre. It demonstrates that the first person is precise and accurate.
- In the second image, all of the arrows hit in the same spot but not in the centre. In this case, the second person is precise but inaccurate.
- On the other hand, the third person is neither precise nor accurate.