
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoChemistry in day to day life:
In our day to day life, we use foods, medicines, cosmetics, dress materials and gold covering ornaments. Of course, they may change in nature and applications, but all these are associated with chemistry.

Applied chemistry in everyday life
We face a lot of difficulties in different means to lead our day to day life. Chemists develop new concepts and principles as a result of such challenges. For example, when people suffered from diseases, new chemical compounds were prepared and used as drugs. Chemists also developed new techniques to diagnose these diseases.
Applied chemistry:
When farmers suffered from low crop yield and pest-related issues in a crop field, chemists produced new chemical fertilisers and pesticides to fight those issues. Thus, chemical principles and methods are applied to several areas to attain specific results or solve real-world problems. This is called applied chemistry.

Applied chemistry in various fields
In this lesson, let us discuss different parts of applied chemistry and their importance.
Nanochemistry:
We know that the size and shape of a material influence its properties. For example, scientists discovered that materials that have a size of about \(1/1,000,000,000\) meters exhibit unique properties. Then they began creating such kinds of materials and studied the effect of size on characteristics. Thus, a new part of chemistry called 'Nanochemistry' was invented.

Nanochemistry laboratory
- Nanochemistry is a part of nanotechnology concerned with the chemical applications of nanomaterials.
- It involves synthesising and manipulating materials at the atomic and molecular levels and studying their physical and chemical properties.
Size of Nanoparticles:
The term Nano is obtained from the Greek word 'Nanos,' representing the millionth part of a unit.
For example, \(1\) nanometer = \(1/1,000,000,000,000,000\) meter.
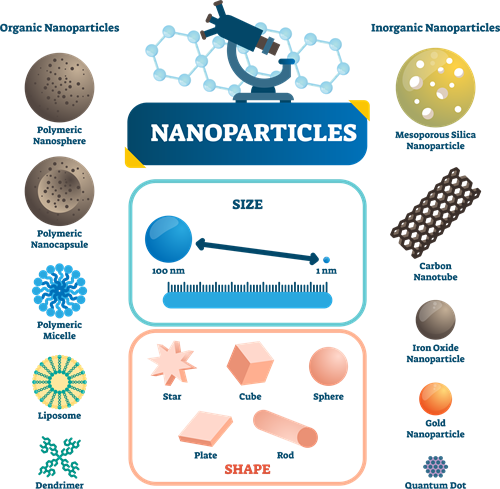
Can you imagine how tiny a nanoparticle is?
The examples given below may help you to demonstrate how small the nanoscale is.
- One nanometer (nm) equals \(10^-{^9}\) or \(0.000000001\) meters.
- A nanometer and a meter are equivalent to the size difference between a golf ball and the Earth.
- Our nails grow at a rate of \(1\) nm per second.
- The virus that usually causes the common cold has a diameter of \(30\) nm.
- A cell membrane is approximately \(9\) nm wide.
- The expanse of the DNA double helix is \(2\) nm.
- One hydrogen atom has a diameter of about \(0.2\) nm.