PDF chapter test TRY NOW
Gay Lussac's law states that the volumes of the reacting gases bear a simple ratio, which is applied to the product when the product is also in the gaseous state. It also states that all the volumes are calculated under identical conditions of temperature and pressure.
The following example helps you to illustrate this law:
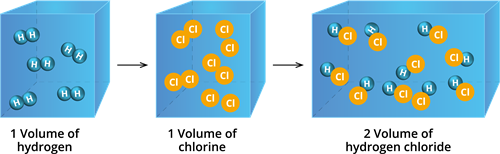
Reaction of hydrogen with chlorine
It has been experimentally observed that one volume of hydrogen reacts with one volume of chlorine to form two volumes of hydrogen chloride, as shown in the figure.
Gases have a volume ratio of \(2\) : \(1\) : \(2\), which is a basic whole-number ratio.
Quantum numbers
We have already studied about shells or orbitals in lower classes.
Quantum numbers are the numbers that designate and distinguish different atomic orbitals and electrons in an atom.
| Quantum numbers | Symbol | Information conveyed |
Principal quantum number | n | Orbital size and energy level |
Azimuthal quantum number | l | Orbital shape Sublevel/subshell |
Magnetic quantum number | m | Orientation of orbitals |
Spin quantum number | s | Electron spin direction |
Points to remember:
- The structure of an atom has been proposed by various scientists, as shown below:
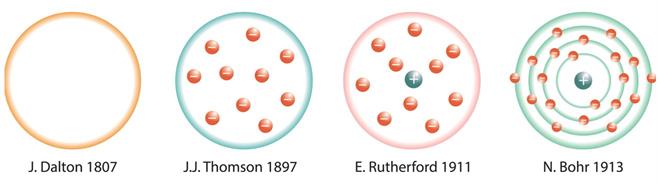
Atomic models
- The sub-atomic particles of an atom are:
Subatomic particles
- The shells or energy levels of an atom are represented by K, L, M, N, etc.
- The combining capacity of an atom is called valency.
- Atomic number = Number of protons in the nucleus.
- Nucleons are protons and neutrons in the nucleus.
- Mass number = Number of nucleons
- Isotopes are atoms with the same atomic number but different mass numbers.
- Isobars are atoms of the same mass number but different atomic numbers.
- Isotones are atoms with the same number of neutrons but different atomic numbers and masses.
- Quantum numbers designate and distinguish different atomic orbitals and electrons in an atom.
