PDF chapter test TRY NOW
The temperature at which the pressure and volume of a gas theoretically reach zero is called absolute zero.
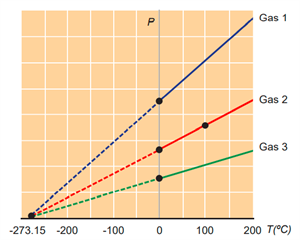
Variation of pressure with temperature
At a temperature of \(–273.15\) \(°C\), the pressure extrapolates to zero for all gases. It is called n as absolute zero or \(0\) \(K\). Some baseline temperatures in the three temperature scales are shown below.
Temperature | Kelvins (\(K\)) | Degree celcius (\(°C\)) | Degrees Fahrenheit (\(°F\)) |
| Boiling point of water | \(373.1\) | \(100\) | \(212\) |
| Melting point of ice | \(273.1\) | \(0\) | \(32\) |
| Absolute zero | \(0\) | \(-273\) | \(460\) |
We can convert the values from one unit to another unit by using the formula,
a) Convert \(30\)\(°C\) to \(Kelvin\)
Solution:
b) Convert \(240\) \(K\) to \(°C\)
