
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoChromatography is a significant biophysical technique for qualitative and quantitative analysis that allows for the isolation, identification, and purification of the components of a mixture.

Colours
The technique of chromatography is being used to separate solutes that are dissolved in the same solvent.
Let us know about the two important terms:
- Adsorption is a chemical reaction in which the particles of one substance are concentrated entirely on the surface of another substance.
Example:The surface of a chalk stick dipped in ink retains the ink's colour due to the adsorption of coloured molecules.
- The process by which a substance is uniformly distributed throughout the bulk of another substance is known as absorption.
Example:When a chalk stick is dipped in ink, the ink's solvent penetrates deeper into the stick due to absorption.
There are many types of chromatography techniques.
Paper chromatography:
Paper chromatography is a method for separating chemical mixtures into their constituent compounds. The paper used in this process is made of cellulose.
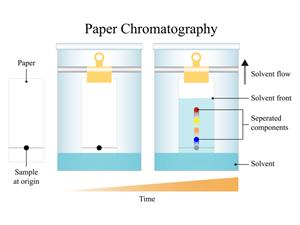
Paper chromatography
Paper chromatography is divided into two phases: A mobile phase and a stationary phase. Paper is the stationary phase, and the solvent is the mobile phase.
Factors determining the components' ability to travel with the solvent are:
- The sample molecule's polarity. Non-polar components move faster than polar components.
- The attraction of the sample molecule to the solvent or combination of solvents.
- The silica and the sample are attracted to each other.

Separation through chromatography
The compound mixture passes through the stationary phase with the mobile phase and separates based on the degree of adhesion (on the paper) of each component in the sample or compound mixture.
Lets us now check if the black ink stain is only a single colour:
Example:

Placing a small drop of water-soluble ink.
Step 1: Take a thin strip of filter paper.
Step 2: With a pencil, draw a line approximately \(3\) cm above the lower edge.
Step 3: Place a small drop of water-soluble ink (from a sketch pen or fountain pen) in the middle of the line. Allow time for it to dry.
Step 4: Lower the filter paper into a pan, bottle, beaker, or test tube filled with water until the ink drop on the paper is just above the line, then leave it alone.
Step 5: As the water rises to the top of the filter paper, keep an eye on it.

Significant rise in the colour.
Observation: There is a significant rise in the colour from its initial point. Water is the solvent in our pigment, and the dye is soluble in it. The dye particles are carried along by the rising water on the filter paper.
Result: A dye is usually a combination of two or more colours. The coloured part that is more soluble in the water rises faster, separating the colours.
Applications:
- Colours in a dye can be separated using this method.
- Pigments derived from natural substances can be separated.
Chromatography