
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoPure substances are those that are completely made up of one form of particle. Physical processes cannot distinguish pure substances from other forms of matter.
Example:
Salt, sugar, oxygen, copper, iron etc.,
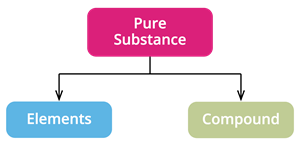
Based on the kind of atom, we classify pure substances as
- Elements and
- Compounds
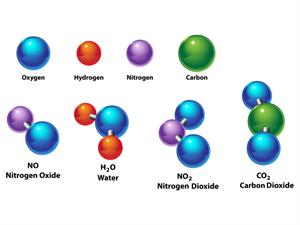
Compound is a form of matter created by combining two or more elements in a specific mass ratio. To decompose a compound into its constituent components chemical methods are used.
Water\(H_2O\), Oxygen \(O_2\), Nitrogen dioxide \(NO_2\), Salt \(NaCl\), and so on.
Water\(H_2O\), Oxygen \(O_2\), Nitrogen dioxide \(NO_2\), Salt \(NaCl\), and so on.
Elements are the fundamental substances that cannot be separated into simpler substances by any chemical methods. All the elements in the periodic table comes under this category such as helium, hydrogen, oxygen, iron, etc.
In \(1661\), Robert Boyle became the first scientist to use the term element.

Robert Boyle
A French chemist named Antoine Laurent Lavoisier \(1743\) - \(1794\) was the first to define an experimentally useful description of an element.

Antoine Laurent Lavoisier
An element, according to him, is a fundamental type of matter that cannot be broken down into simpler substances by chemical reactions.
- Metals
- Non-metals
- Metalloids
Let us now identify some of the elements and compound of a pure substance:
Pure Substance | Element /Compound | Reason why it is compound or element |
| Water \(H_2O\) | Compound | Can be separated as Water = Hydrogen + Oxygen |
| Lead \(Pb\) | Element | Cannot be separated |
| Phosphorous \(P_4\) | Element | Cannot be separated |
| Sodium Chloride \(NaCl\) | Compound | Can be separated as Sodium Chloride = Sodium + Chloride |
| Sulphur \(S_8\) | Element | Cannot be separated |