
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoElectrolysis:
When an electric current is used for decomposing a substance, it is called electrolysis.
Let us perform an activity (electrolysis of water) to understand the process of decomposition (single reactant (water) breaks down to give simpler products (hydrogen and oxygen)).
Activity \(1.7\): Electrolysis of water.
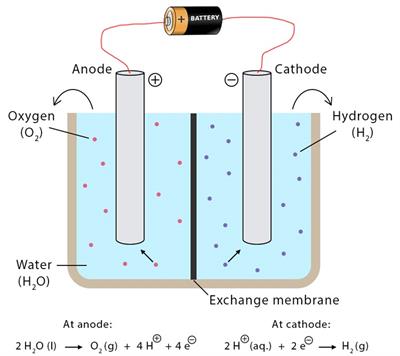
The process in which water is decomposed by passing electricity is called the electrolysis of water.
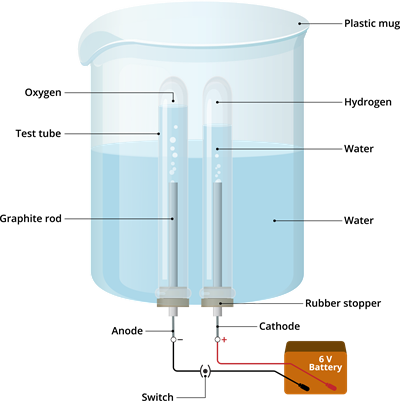
Materials required:
-Plastic mug
-Rubber stopper
-Carbon electrodes (anode & cathode)
-Battery
-Water
-Dilute sulphuric acid and
-Test tube
Experimental procedure:
Step 1: Take the plastic mug and drill two holes at its base.
Step 2: Fit the two rubber stoppers in these holes.
Step 3: Insert the carbon electrodes (anode & cathode) into these rubber stoppers.
Step 4: Connect these carbon electrodes to \(6\)-volt battery.
Step 5: Now, fill the plastic mug with water up to a level such that the carbon electrodes get immersed.
Step 6: Add few drops of sulphuric acid to this water.
Step 7: Take two test tubes to fill them with water; after that, invert these test tubes to the carbon electrodes.
Step 8: Now, switch on the current from the \(6\)-volt battery.
Step 9: Leave the apparatus undisturbed for some time.
Observation:
- There will be the formation of bubbles at both the electrodes in the test tubes (which means gas is forming or liberating from water)
- Due to the formation of bubbles, water starts moving in the test tube.
Step 10: Once these test tubes are filled with the gases, carefully remove them from the mug.
Important!
The teacher must carefully carry out this step.
Step 11: Take a glowing candle near the mouth of the test tubes to check which gas is present in them.
Electrolysis of water
Result:
- When we bring a glowing candle close to the mouth of one of the test tubes, the gas in the test tube takes fire and burns with a pop sound, showing the presence of hydrogen in the test tube.
- When we bring a burning candle closer to the mouth of another test tube, the candle starts to burn brightly, showing that the test tube contains oxygen.
- The gas collected at the anode (negative) is oxygen, and the gas collected at the cathode (positive) is hydrogen.
The volume of the gas obtained in both the test tubes is not the same. The hydrogen gas volume is twice the oxygen gas volume contained in the other test tube.
Decomposition reaction:
\(2H_2O\)(l) \(2H_2\)(g) + \(O_2\)(g)
(Water) (Hydrogen) + (Oxygen)
Conclusion:
In this reaction, you can notice that a single reactant breaks down to give simpler products, known as a decomposition reaction. Here water decomposes to form hydrogen and oxygen under suitable conditions. In this case, the electricity is the suitable condition under which the water gets decomposed.
Reference:
https://www.chemistrylearner.com/electrolysis-of-water.html