
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoLet us discuss some additional examples of combination reaction.
(i). Burning of coal

Coal burning
Coal burns in the air to form carbon dioxide gas.
(ii). Formation of water from \(H_2\)(g) and \(O_2\)(g)

Water formation reaction
\(2H_2\)(g) + \(O_2\)(g) → \(2H_2O\)(l)
In the presence of oxygen, hydrogen burns to produce water in the form of steam. Upon cooling, it becomes liquid.
Combustion reactions: Burning of coal and formation of water are examples of combination reaction.
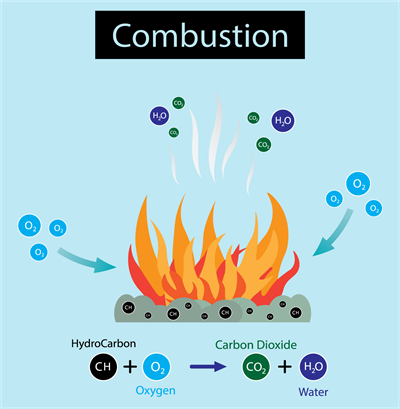
In simple expression, we can state that when two or more substances react to produce a single product, the reactions are termed combination reaction.
Additional examples of exothermic reactions are-
(i). Burning of natural gas
Natural gas burning is an example of an exothermic reaction.
The burning of natural gas is a combination reaction. Natural gas mainly consists of methane.
\(CH_4\)(g) + \(2O_2\)(g) → \(CO_2\)(g) + \(2H_2O\)(g)
In combustion reactions, the products are always carbon dioxide and water. In a combination reaction, two reactants react to form a product.
(ii). Are you aware that respiration is an exothermic process?
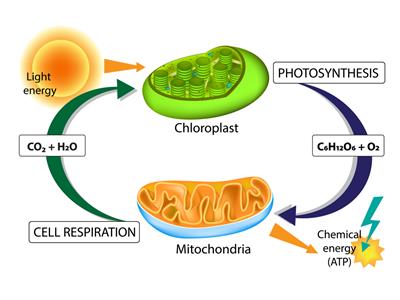
Respiration
We all know that we want the energy to stay alive. We take this energy from the food we eat. Through digestion, food is broken down into simpler compounds. For example, rice, potatoes and bread include carbohydrates. These carbohydrates are cut down to form glucose. Glucose reacts with oxygen in the cells of our body and provides energy. The particular name of this reaction is respiration.
\(C_6H_12O_6\)(aq) + \(6O_2\)(aq) → \(6CO_2\)(aq) + \(6H_2O\)(l) + Energy
(Glucose)
(Glucose)
(iii). The decomposition of vegetable components into compost is an exothermic process.

Vegetable decomposition
Recognize the type of reaction in Activity \(1.1\) that forms heat and a single product.
In the activity \(1.1\), we noticed that a large amount of heat is evolved. That heat makes the reaction mixture warm. Reactions in which heat is delivered along with the formation of products are named exothermic reactions. So the activity \(1.1\) is an example of exothermic reactions.