
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
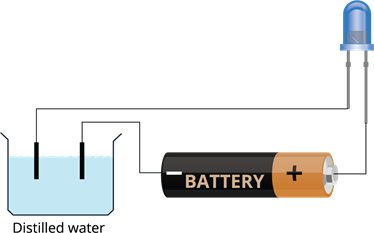
Book Free DemoElectrical conductivity of distilled water:
- Take distilled water in a plastic container.
- Use LED instead of bulb since LED glows even when a weak electric current flows through it.
- Make a battery (a couple of cells) and connect its positive terminal to the longer lead of the LED. Put the negative terminal of the battery into the container that contains distilled water.
- Put the shorter lead of the LED too into the water using a wire.
- Make sure that both ends in the water do not touch and keep at least \(1\) cm distance.
Experiment setup
In this experiment, we can see that the LED does not glow that means the circuit is not closed; hence, the electricity is not passing through the distilled water to complete the circuit. Thus, distilled water is a poor conductor of electricity. It has no salt or impurities in it.
Magnetic needle deflection:
As we studied earlier, current produces magnetic field. So,we can use magnetic needle deflection to detect the conductivity of a material even when a weak current is passing through it.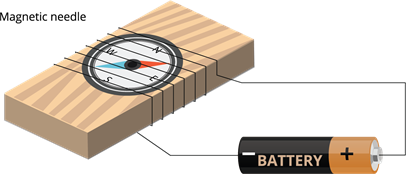
Experiment setup
- Take a tray of an empty matchbox.
- Place a small compass needle inside the box.
- Wrap an electric wire around the box a few times.
- Connect the free ends of the wire to a battery.
- Now, we can see the deflection in the needle. Thus, our tester of two free ends is ready.
If we use this experiment for lemon juice or vinegar, or water, it will show the deflection in the magnetic needle, which means the liquids are good conductors.