PDF chapter test TRY NOW
We have studied that every tiny particle of this universe is made up of matter.
- Pure substances
- Impure substances
Classification of Matters:
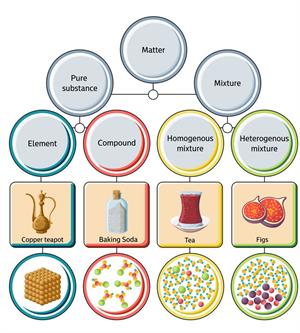
Classification of matters
What is a pure substance?
The substances which are made up of only one kind of particles are known as pure substances. These are substances that cannot be separated into any other kinds of matter by any physical process.

Red balloons
The above picture containing red balloons of the same size, shape and weight etc., is an example of a pure substance, as we cannot separate these into two kinds of matter.
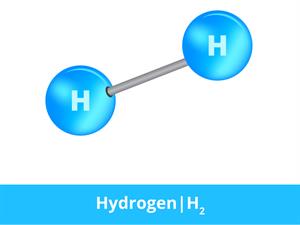
Hydrogen molecule
In hydrogen gas, the substance is made of the same atoms. Thus, two hydrogen atoms bond together and form a hydrogen molecule which is in a gaseous state.
The other examples of pure substances are sugar, salt, diamond, etc. Crystals, in general, are known as pure substances.


Sugar

Oxygen

Pure gold (\(24\) carat)
What are impure substances?
The combination of more than one kind of substance is known as an impure substance or mixture. Mixtures can be separated by physical process.

Fruit salad
In a fruit salad, most of \(4\) to \(5\) kinds of fruits are mixed together. It is an example of a mixture, as we can separate them by handpicking method into separate fruits.

Lemon juice with honey and water
In fresh juice, fruit is mixed with water and sugar. There are three different kinds of substances present in it, so it is called a mixture.
The mixture is further classified into two types based on the property of substances they are made up of.
The mixture is further classified into two types based on the property of substances they are made up of.
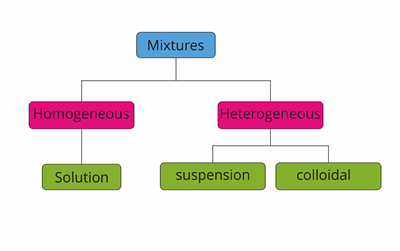
Classification of mixtures
Example:
Seawater, petroleum, air, muddy water, milk and cereals.
Tyndall effect is a light scattering effect in colloidal dispersion that appears to show no light in a true solution. This effect is used to decide whether a solution or a colloid is present in a mixture.

Let us know some facts about the Tyndall Effect.
