
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoLet us check if we can separate the components of air.
Air is a homogeneous mixture. We cannot use filtration, centrifugation and chromatography for this method of separation.
Air is a homogeneous mixture that can be fractionally distilled to isolate its constituents.
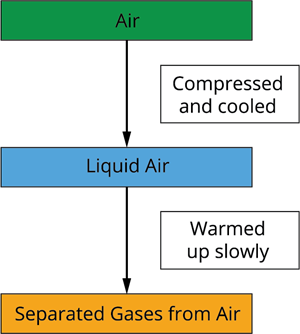
Separation of components
Process flow:
The boiling point of components of air are as follows

Air components separation
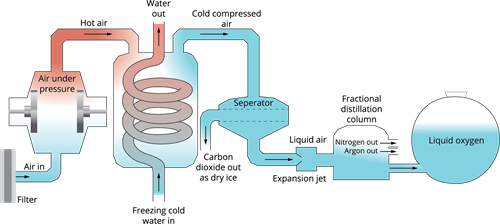
The process of separation of oxygen from the air
Step 1: To get oxygen gas from the air, we must first separate all other gases present. The air is compressed by increasing the pressure and then cooled by lowering the temperature to obtain liquid air.
Step 2: This liquid air is allowed to slowly warm up in a fractional distillation column, where gases are divided at different heights based on their boiling points.
Result: The required gases from the air is separated accordingly using this method.