PDF chapter test TRY NOW
Nitrogen cycle is a bio-geo-chemical process-atmospheric air contains \(78%\) of nitrogen (the abundant nitrogen source). Nitrogen is present in proteins and nucleic acids (amino acids) essential to life.
With the help of microbial transformations, plants use nitrogen, which in turn rotates to the environment.
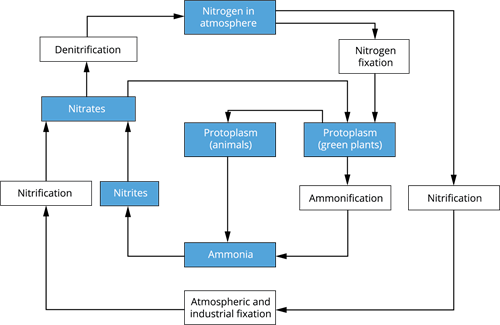
The process of the nitrogen cycle are
- Nitrogen fixation
- Nitrification
- Nitrogen assimilation
- Ammonification
- Denitrification
Nitrogen fixation ( converts to ):
Nitrogen is present in the atmosphere as dinitrogen gas (); in this form, it is mostly inaccessible to most organisms. When nitrogen is converted from dinitrogen gas to ammonia (), it is used by primary producers in our ecosystem. This process is known as nitrogen fixation. There are three types
- Biological process
- Natural process
- Synthetic process
1. Biological process:
In this step, the nitrogen gas present in the atmosphere is converted biologically by prokaryotes. This conversion is carried out by bacteria (non-symbiotic) and blue-green algae, where they fix almost about \(90%\) of nitrogen and are called natural nitrogen fixers.
Example:
Bacteria such as cyanobacteria (nitrogen fixers in aquatic environments), azotobacter (nitrogen-fixing bacteria in the soil), and clostridium (nitrogen-fixing bacteria in the soil).
 |
The symbiotic nitrogen-fixing bacteria invade host plants' root hairs, where they multiply and stimulate the formation of root nodules. The bacteria convert free nitrogen to ammonia within the nodules, which the host plant utilises for its development.
The presence of nitrogenase enzyme helps these bacteria for the conversion process of atmospheric nitrogen.
Example:
Rhizobium (leguminous plants) and Frankia (certain dicotyledonous species).
2. Natural process:
A minimal amount of free nitrogen is fixed by abiotic means such as electrical equipment, radiation, lightning, etc.
3. Synthetic process:
Haber-Bosch process:
In this process, ammonia is produced from atmospheric air and hydrogen under high pressure 200-400 atmosphere and temperature in the presence of a catalyst based on iron supported by . This is an industrial method for ammonia production.
Nitrification ( converts to ):
The ammonia gas is not used directly by plants as it is toxic, so further conversion occurs. The first step is the conversion of ammonia into nitrites () by the oxidation process of the bacteria Nitrosomonas. Then, the second step is the conversion of nitrites () to nitrates() by Nitrobacter.
Step 1:
Step 2:
Assimilation (Incorporation of , ):
The nitrates and ammonia are formed taken up by plants via roots and is incorporated into their tissues. After this, animals consume these producers, thereby converting them into their body compounds. It is through this process by which nitrogen enters our food chain.
The nitrates and ammonia are formed taken up by plants via roots and is incorporated into their tissues. After this, animals consume these producers, thereby converting them into their body compounds. It is through this process by which nitrogen enters our food chain.
Ammonification (formation of by decomposition):
The decomposition of dead plants and animals by bacteria and fungi present in the soil helps fix the ammonium back to the soil. The decomposition process produces ammonia.
Denitrification ( converts to ):
The process of releasing the nitrogen back to the atmosphere is denitrification. The bacterium is active in water-logged anaerobic soils such as Clostridium and Pseudomonas, which helps in this process of denitrification.These bacteria deplete the soil's nitrates and form free atmospheric nitrogen.
The process of releasing the nitrogen back to the atmosphere is denitrification. The bacterium is active in water-logged anaerobic soils such as Clostridium and Pseudomonas, which helps in this process of denitrification.These bacteria deplete the soil's nitrates and form free atmospheric nitrogen.
Reference:
Stdout, CC BY-SA 3.0 <https://creativecommons.org/licenses/by-sa/3.0>, via Wikimedia Commons
<a href="https://www.vecteezy.com/free-vector/nitrogen-cycle">Nitrogen Cycle Vectors by Vecteezy</a>
