PDF chapter test TRY NOW
The circulation of carbon from the atmosphere to the earth is known as the carbon cycle. This is a biogeochemical process.
The amount of carbon present in the atmosphere is \(0.04\%\). This is also released by the respiration process and on the complete combustion process. Carbon occurs in its elemental forms as graphite and diamond in nature.

The following is the process involved in the carbon cycle
- Plants take in atmospheric for photosynthesis.
- Animals feed on primary producers, and carbon is accumulated in them.
- The carbon is released back to the atmosphere on the decomposition of dead plants and animals.
- Some amount of carbon is retained in the soil and becomes fossil fuel.
- The combustion of fossil fuels by human activities releases carbon back to the atmosphere.
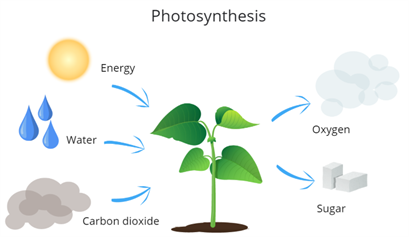 |
The intake of from the atmosphere for preparing food with the help of sunlight is known as photosynthesis.
The carbon is passed from producers to consumers in the ecosystem from this process.
Fact: \(18.5\%\) of carbon is present in the human body.
Carbon sink: The intake of is more compared to the release of .
 |
The plants, soil and ocean act as a natural carbon sink. We know the shells of marine organisms are made of calcium carbonates. So when the marine organisms die, they get accumulated in the seafloor, and decomposition takes place.
Apart from the natural release, there is also an additional amount of which is released into the atmosphere due to modernisation techniques, burning, industrialisation etc., which can alter the carbon cycle. If the carbon cycle is altered, it will have a drastic effect on earth, which concerns the life on earth and other conditions such as global warming, climate change, etc.
Reference:
Harry C, CC BY-SA 3.0 <https://creativecommons.org/licenses/by-sa/3.0>, via Wikimedia Commons
