PDF chapter test TRY NOW
Alpha decay:
Alpha decay is a nuclear reaction in which an unstable parent nucleus emits an alpha particle and forms a stable daughter nucleus.
The decay of Uranium (\(U^{238}\)) to Thorium (\(Th^{234}\)) with the emission of an alpha particle.
As the parent nucleus emits an \(α -\ particle\) during the decay, it is evident that the mass number of the daughter nucleus reduces by four and the atomic number decreases by two.
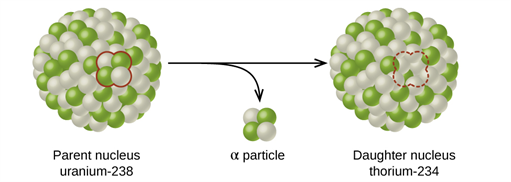
Alpha decay process
Beta decay:
Beta decay is a nuclear reaction in which an unstable parent nucleus emits a beta particle and forms a stable daughter nucleus.
Beta decay of phosphorous is a good example.
\(\)
\(\)
In \(β-\ decay\), the atomic number increases by one, but there is no change in the mass number of the daughter nucleus.
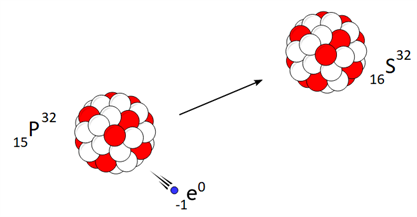
Beta decay process
Important!
NOTE: The atomic number of the resulting nucleus identifies the product nucleus in a nuclear reaction.
Gamma decay:
In gamma decay, only the energy level of the radioactive nucleus changes, but the atomic number and mass number remains the same.
Gamma decay process
In radioactive decay, both \(α\)-decay and \(β\)-decay never take place together in a single process. Either \(α\)-decay or \(β\)-decay occurs along with the emission of \(γ\) particles.
Reference:
https://upload.wikimedia.org/wikipedia/commons/3/37/Gamma_Decay01.svg
