PDF chapter test TRY NOW
Beta (\(β\)) rays are the electrons (\(_{-1}e^{0}\)), one of the fundamental elementary particles in an atom. They are negatively charged particles, having a charge of \(–e\).
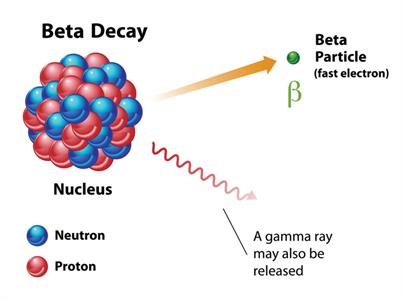
Ejection of a beta particle
Properties of beta particles:
1. The beta particles have comparatively low ionising power than the alpha particles.
2. The penetrating power of beta particles is greater than that of \(α\) particles. The particles can penetrate through a thin metal foil.
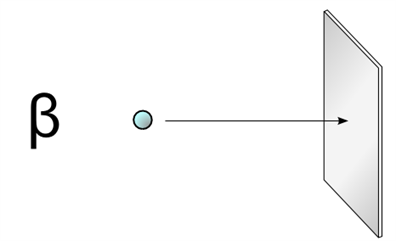
Beta particle hitting a metal
3. According to Fleming’s left-hand rule, both the electric and magnetic fields deflect the beta particles. But the deflection is opposite to that of alpha rays.
4. The speed of beta rays can reach up to \(9/10\) times the speed of light.
Gamma rays:
Gamma (\(γ\)) rays are electromagnetic waves consisting of photons.

Gamma rays
Properties of gamma particles:
1. Gamma particles are chargeless or neutral particles with very low ionising power.
2. Gamma rays have a very high penetrating power greater than that of \(β\ rays\), such that they can penetrate through thick metal blocks.
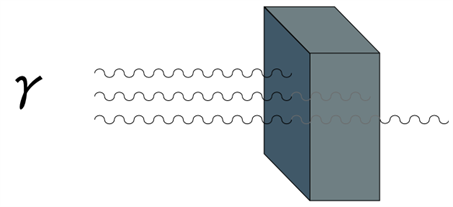
Gamma particle hitting a metal block
3. According to Fleming’s left-hand rule, the particles are not deflected by electric and magnetic fields.
4. Gamma rays can travel with the speed of light.
Radioactive displacement law:
In \(1913\), Soddy and Fajan defined the displacement equations to regulate the daughter nucleus formed during alpha and beta decay.(i) When a radioactive element emits an alpha particle, a daughter nucleus is formed whose mass number is less by \(4\) units, and the atomic number is less by \(2\) units than the mass number and the atomic number of the parent nucleus.
(ii) When a radioactive element emits a beta particle, a daughter nucleus is formed whose mass number is the same, and the atomic number is more by \(1\) unit than the atomic number of the parent nucleus.
Reference:
https://upload.wikimedia.org/wikipedia/commons/7/7b/Gamma_radiation_%28alternative%29.svg
https://upload.wikimedia.org/wikipedia/commons/5/52/Alfa_beta_gamma_neutron_radiation_M1.PNG
