PDF chapter test TRY NOW
Sun is the important and primary source of thermal energy for all living organisms. Thermal energy is the cause, and temperature is the effect. All living organisms require a particular temperature for their survival.
All of us have common knowledge about thermal energy and temperature. But, in this chapter, we will discuss about thermal energy and temperature in a scientific manner. We shall also discuss how thermal energy is transferred and the effects of thermal energy.
Temperature
The measurement of the hotness or coldness of a substance is known as its temperature. It determines the direction of the flow of heat when two bodies are placed in contact.
Units of Temperature:
Three units are used to measure the temperature:
- Degree Celsius
- Fahrenheit and
- Kelvin
Degree Celsius:
Celsius is written as °\(C\) and read as the degree.
For example, \(100\)°\(C\) is read as a 'hundred degree Celsius'. Celsius is also called Centigrade.
Thermometer - Celsius scale
Fahrenheit:
Fahrenheit is written as °\(F\). F
For example, \(100\)°\(F\) is read as a 'hundred degree Fahrenheit'.

Thermometer - Fahrenheit scale
Kelvin:
Kelvin is written as K.
For example, \(100\)\(K\) is read as a 'hundred Kelvin'. SI unit of temperature is Kelvin.
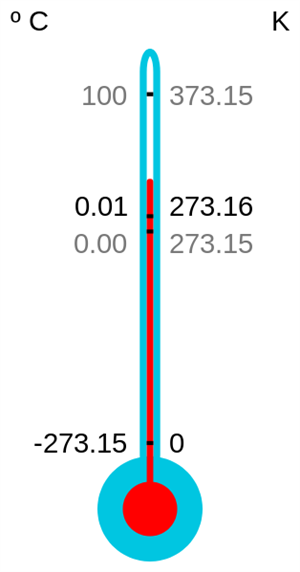
Thermometer - Kelvin scale
Measuring Temperature:
The temperature of the substance is the approximation of the average kinetic energy of the substance. Higher temperature means the molecules inside the substance are moving rapidly; similarly, lower temperature means that the molecules are moving in a slow phase
But the question is, how to measure the temperature?
The movement of the molecules is directly proportional to the temperature. Since the molecules in any substance are very small to compute, it is really difficult to measure the movement of the molecules (Kinetic energy). So we need to look at indirect ways to measure this molecule's movement (Kinetic energy).
Solids, Liquids and Gases are affected by heat. It expands when heated and contracts when cooled. This principle is used to measure the temperature.
The thermometer is used to measure the temperature.
In a thermometer, when liquid gets heat, it expands, and when cooled down, it contracts. The expansion and contraction of liquid is used to measure temperature.
Reference:
https://pixabay.com/fr/vectors/thermom%C3%A8tre-temp%C3%A9rature-1134182/
https://upload.wikimedia.org/wikipedia/commons/d/d2/Barthermometer_Fahrenheit%2BCelsius.jpg
https://commons.wikimedia.org/wiki/File:CelsiusKelvin.svg
https://upload.wikimedia.org/wikipedia/commons/d/d2/Barthermometer_Fahrenheit%2BCelsius.jpg
https://commons.wikimedia.org/wiki/File:CelsiusKelvin.svg
