PDF chapter test TRY NOW
A reversible reaction is a reaction in which the products can be converted back to the reactants. A reversible reaction is represented by a pair of arrows pointing in opposite directions.
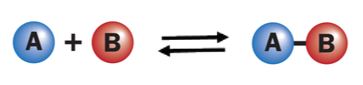
Reversible reaction
Here, the compound ‘AB’ decomposes to form‘ A’ and ‘B’. It is a forward reaction. The products combine to form ‘AB' as soon as they are formed. It is a backward reaction. Hence, the reaction occurs in both directions.
Do you think that in the process mentioned above, no products are formed? If you think so, you are mistaken. Because, even if the reaction occurs in both directions, the rates (speed) of the reactions are not equal initially. Consider the decomposition of phosphorous pentachloride into phosphorous trichloride and chlorine in the following reaction.
In the forward reaction, the decomposition of \(PCl_5\) occurs, and in the backward reaction, the combination of \(PCl_3\) and \(Cl_2\) occurs. The forward reaction is initially faster than the backward reaction. After a while, the speed of both reactions become equal.
Therefore, as the reaction is reversed, \(PCl_5\) cannot completely convert into the products. The reaction is reversible. The reaction appears to be in equilibrium based on actual measurements. However, the amount of \(PCl_5\) is more than the amounts of \(PCl_3\) and \(Cl_2\).
Thus, in a reversible reaction, new products can be obtained by periodically removing one of the products or periodically adding the reactants.
Hydrogen peroxide decomposes into water and oxygen when applied to a wound. As it forms, gaseous oxygen bubbles go away, preventing the formation of \(H_2O_2\).
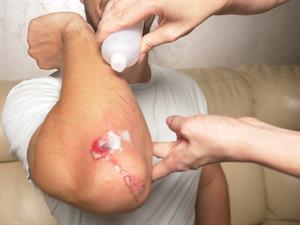
Hydrogen peroxide on a wound
Irreversible reaction:
The reaction that cannot be reversed is called an irreversible reaction.
Irreversible reactions are unidirectional, meaning they only occur in one direction. Consider the process of coal burning, which produces carbon dioxide and water.
\(C_{(s)} + O_{2(g)} → CO_{2(g)} + Heat\)
Solid coal is burned with oxygen in this reaction, resulting in the production of carbon dioxide gas and water. Since the product is a gas, it escapes the reaction container as soon as it is created. Decomposition of a gas into a solid is extremely difficult. As a result, in this case, a reverse reaction is not possible. Hence, it is an irreversible reaction.
Differences between reversible and irreversible reactions:
Reversible reaction | Irreversible reaction |
Under suitable conditions, it can be reversed. | It cannot be reversed. |
Both forward and backward reactions occur simultaneously. | It is unidirectional. This reaction occurs only in the forward direction. |
It attains equilibrium. | It does not attain equilibrium. |
It is not possible to convert reactants completely into products. | It is possible to convert reactants completely into products. |
It is a slow process. | It is fast. |
| This process results in a temporary change. | This process results in a permanent change. |
This reaction is represented by equilibrium arrow, i.e., ' ' | This reaction is represented by normal arrow, i.e., '' |
