PDF chapter test TRY NOW
When two compounds react, if their ions are interchanged, the reaction is called a double displacement reaction.
Ions with identical charges can only be interchanged, which means that a cation can be replaced by another cation. This reaction is also called ‘Metathesis reaction’.
Many displacement reactions occur between the ionic compounds that are dissolved in water. A double displacement reaction is shown schematically as follows:
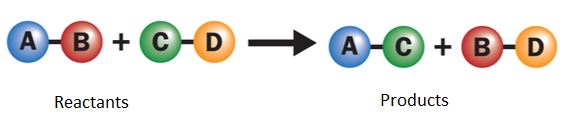
Double displacement reaction
Atoms from two different compounds swap places in a double displacement reaction. The two reactants and products are different compounds. For example:
\(2NaOH+CuSO_4→Cu(OH)_2+Na_2SO_4\)
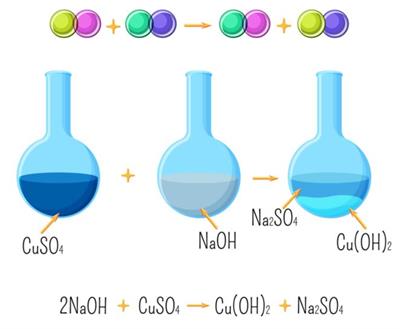
Double displacement reaction
In a double displacement reaction, one of the products must be either a precipitate or water. In this way, double displacement reaction is further classified as follows:
(i) Precipitation reactions
(ii) Neutralization reactions
Precipitation reactions:Since one of the products is an insoluble compound, the reaction is called a precipitation reaction.
When aqueous solutions of two compounds are mixed, if they react to form an insoluble compound and a soluble compound, it is called a precipitation reaction.
Example:
Mixing the clear aqueous solutions of potassium iodide with lead (II) nitrate is an example of a double displacement reaction.
\(Pb(NO_3)_{2(aq)}+2KI_{(aq)}→PbI_{2(s)}↓+2KNO_{3(aq)}\)
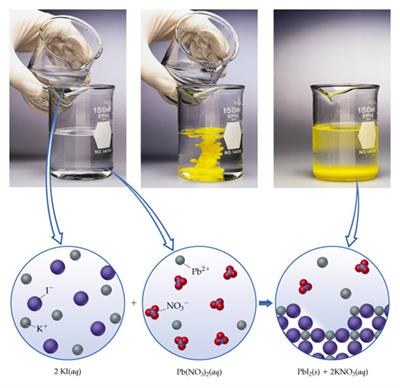
Precipitation of \(PbI_2\)
In this reaction, potassium and lead displace or replace one another and form a yellow precipitate of lead (II) iodide.
Neutralization reactions:
The reaction between an acid and a base result in the formation of salt and water is called a neutralisation reaction. This is another type of displacement reaction in which both acid and base neutralise each other.
Example: \(1\)
A common neutralisation reaction is the reaction of sodium hydroxide with hydrochloric acid. Here, sodium replaces hydrogen from hydrochloric acid, forming sodium chloride (neutral soluble salt).
\(NaOH_{(aq)} + HCl_{(aq)} → NaCl_{(aq)} + H_2O_{(l)}\)

Neutralisation reaction
Example: \(2\)
The reaction of ammonium hydroxide with nitric acid forms ammonium nitrate and water.
\(HNO_{3(aq)}+NH_4OH_{(aq)}→NH_4NO_{3(aq)}+H_2O_{(l)}\)
Reference:
https://th.bing.com/th/id/R.d179796511bf2be8ba3a9921dc8401f2?rik=h%2fCCtE0YWYMeLA&riu=http%3a%2f%2fchemwiki.ucdavis.edu%2f%40api%2fdeki%2ffiles%2f76%2fsynthsis_reaction2_(1).png%3frevision%3d2&ehk=%2ba3PCRQ4X2nOyk6zwdzaCv%2f%2f0A%2bF0ddUZvOjC5GfDmg%3d&risl=&pid=ImgRaw&r=0
https://ecampusontario.pressbooks.pub/app/uploads/sites/372/2019/01/acid_base_reaction.png
