PDF chapter test TRY NOW
A single-displacement reaction, also known as a single replacement reaction or an exchange reaction, occurs when one element in a compound is replaced by another. The following is a schematic representation of a single displacement reaction:
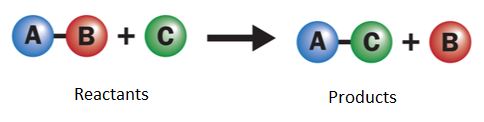
Single displacement reaction
The element 'A' displaces element 'B' from the compound 'BC'; hence, the reaction is referred to as a single displacement reaction.
Example \(1\):
When zinc metal is placed in hydrochloric acid, hydrogen gas is evolved. This implies that hydrogen is displaced by zinc metal, and zinc chloride is formed. This reaction is shown below:
\(Zn_{(s)}+2HCl_{(aq)}→ZnCl_{2(aq)}+H_{2(g)}\)
When an iron nail is placed in an aqueous solution of copper (II) sulphate, the iron displaces copper from its aqueous solution and forms copper deposits over the iron nail.
\(Fe_{(s)}+CuSO_{4(aq)}→FeSO_{4(aq)}+Cu_{(s)}\)
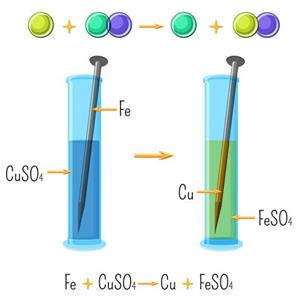
Single displacement reaction

Displacement of copper
It is easy to demonstrate a variety of reactions of this type using various reactant combinations. But none of them occur in practice. This is most easily demonstrated with halogens. Let us consider the following two reactions:
\(2NaCl_{(aq)}+F2_{(g)}→2NaF_{(aq)}+Cl_{2(g)}\)
\(2NaF_{(aq)}+Cl_{2(g)}→2NaCl_{(aq)}+F2_{(g)}\)
Fluorine replaces chlorine in \(NaCl\) in the first reaction. Chlorine displaces fluorine in \(NaF\) in the second reaction. Since fluorine is more active than chlorine and is located higher in the periodic table, the second reaction will not occur. So, in displacement reactions, the activity of the elements and their relative position in the periodic table are the most important factors to determine the feasibility of the reactions. More active elements readily displace less active elements from their aqueous solution.
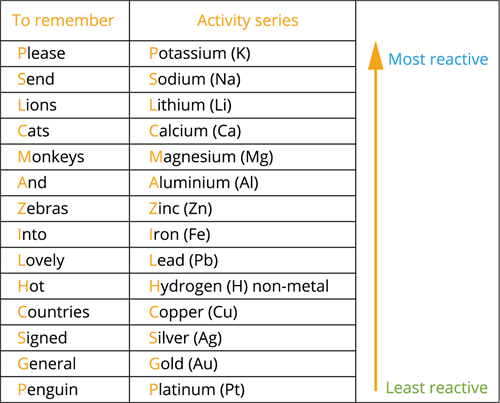
Reactivity series: