PDF chapter test TRY NOW
One-fifth of the atmosphere consists of oxygen by occupying around \(21\%\) of the atmosphere. Oxygen is generally consumed in the process of respiration by all the living things on earth.
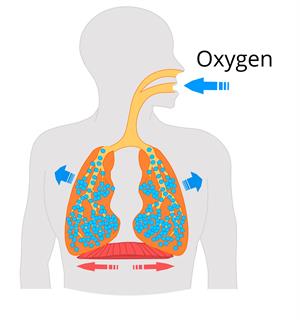
Inhalation of oxygen
Burning a substance in the presence of oxygen along with the release of a large amount of light and heat is known as the burning process.
A continuous supply of fresh air is needed to burn a substance. When the air supply to a burning substance is cut off, the substance will not get enough oxygen and stops the burning process. Hence, oxygen is also needed in the process of burning a candle, paper, kerosene, coal, wood and cooking gas.

Burning of wood
Burning candle experiment:
1. Two lighted candles of the same height are taken and are placed on the table.

Candles placed on the table
2. Now, one of the candles is covered with a transparent glass tumbler.

Covering a candle with glass
3. After some time, the candle covered with a glass tumbler gets extinguished. But, the other one continues to burn.

Extinguished candle
Inference:
In the above experiment, the gas component, which supported the burning of the candle, is known as oxygen.
The candle covered with the glass was extinguished due to the limited amount of oxygen gas present inside the glass tumbler. Only a small quantity of air is present inside the tumbler, and hence only less oxygen is available for the candle to glow. When all the oxygen got exhausted inside the glass, the candle stopped burning.
At the same time, the other candle was burning due to the continued supply of air. The glowing candle may not get extinguished unless it is blown out by a strong wind or any other external force.
