
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free Demo
Ernest Rutherford
- Ernest Rutherford developed Thomson's theory, and he gave a better understanding. He experimented to understand the structure of an atom.
- Rutherford bombarded a very thin layer of a gold sheet with positively charged alpha rays.
- Rutherford found that most of these rays which travel at a great velocity, passed through the gold sheet without any deviation. A few rays are turned back from the sheet. Rutherford considered this as remarkable and miraculous as if a bullet had turned back after colliding with tissue paper.
- He proposed his famous theory based on this experiment.
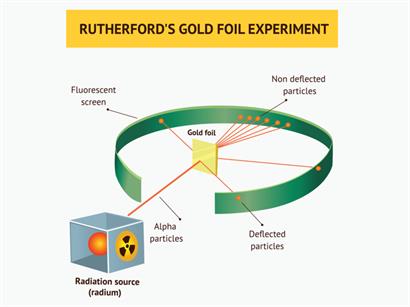
Rutherford's gold foil experiment
Through this observation he concluded the following points,
- Most alpha particles pass through the gold sheet, meaning that the atom mainly consist of free space.
- The part from where the positively charged particles turned back should be positively charged but very small in size as compared to the empty space.
Based on these observations, Rutherford presented his theory of the structure of atoms. In \(1908\), he was given the Nobel prize in chemistry for this theory.
Rutherford proposed that:
- The nucleus is at the centre of the atom and has a positive charge. Most of the mass of the atom is concentrated in the nucleus.
- The electrons whose charge is negative revolve around the nucleus in specific or fixed orbits.
- By comparing the size of the nucleus with the atom's size, the nucleus is very, very small and occupies a small part in the atom.